Rheumatoid arthritis and stroke risk: a systematic review and meta-analysis
- Published
- Accepted
- Received
- Academic Editor
- Faiza Farhan
- Subject Areas
- Epidemiology, Neurology, Rheumatology
- Keywords
- Rheumatoid arthritis, Meta-analysis, Stroke, Systematic review
- Copyright
- © 2026 Yang et al.
- Licence
- This is an open access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, reproduction and adaptation in any medium and for any purpose provided that it is properly attributed. For attribution, the original author(s), title, publication source (PeerJ) and either DOI or URL of the article must be cited.
- Cite this article
- 2026. Rheumatoid arthritis and stroke risk: a systematic review and meta-analysis. PeerJ 14:e20568 https://doi.org/10.7717/peerj.20568
Abstract
Objectives
This meta-analysis aims to evaluate the association between rheumatoid arthritis (RA) and stroke risk. Methods: PubMed, Cochrane Library, and Embase were searched for observational studies published from database inception to October 7, 2025, using medical subject headings (MeSH) and keywords. Random effect models were used to calculate odds ratios (OR) and 95% confidence intervals (CI) for evaluating the associations between RA and stroke risk. All statistical analyses were performed using Stata statistical software version 17.0. The funnel plot, Egger’s test and Begg’s test were used to evaluate publication bias.
Results
This meta-analysis included 12 observational studies with a total of 1,715,001 participants, published between 2003 and 2025. The pooled analysis revealed a significant association between RA and increased stroke risk (OR = 1.35; 95% CI [1.26–1.45]; P = 0.000). Subgroup analysis showed that women with RA had a slightly higher stroke risk than men (OR = 1.60; 95% CI [1.19–2.16]; P = 0.002). Additionally, RA patients aged over 65 were at higher risk of stroke (OR = 1.24; 95% CI [1.02–1.50]; P = 0.032). No significant publication bias was detected, and sensitivity analyses confirmed the robustness of our findings.
Conclusions
This meta-analysis demonstrates that RA is associated with an increased risk of stroke, supporting the recognition of RA as an independent stroke risk factor.
Introduction
Stroke is an acute syndrome and a leading cause of death and disability worldwide (Murphy & Werring, 2020). The Global Burden of Disease (GBD) study reported 11.9 million incident strokes, 93.8 million prevalent strokes, 7.25 million stroke-related deaths, and 160 million Disability-Adjusted Life Years (DALYs) lost to stroke in 2021 (Diseases & Injuries, 2024). Early identification of stroke risk factors is crucial for prevention. Several quantitative meta-analyses have identified key risk factors for stroke, including dementia, sleep insufficiency, migraines, dietary protein intake, cold spell and abnormal body weight (Fan et al., 2023; Kuźma et al., 2018; Li et al., 2016; Zhang et al., 2021; Zhang et al., 2016; Zhao et al., 2024). Recently, rheumatoid arthritis (RA) has emerged as a potential risk factor for stroke due to its association with persistent synovial inflammation and progressive joint destruction (Agca et al., 2017). This chronic inflammation stimulates the release of enzymes, pro-inflammatory mediators, and cytokines, resulting in synovial tissue degradation (Miller, Miller & Malfait, 2014). The increased production of inflammatory cytokines in the joints is a key mechanism connecting rheumatoid arthritis to a higher risk of stroke. These cytokines can enter the bloodstream and increase the production of adhesion molecules and other pro-inflammatory molecules. This leads to monocyte and leukocyte adhesion to the endothelial cells of blood vessels, migration into the vessel walls, and eventually triggering strokes (Libby, 2009; Zaman et al., 2000).
RA is a chronic autoimmune inflammatory disease associated with disability and premature mortality. In 2020, an estimated 17.6 million people worldwide were living with RA, with a global age-standardized prevalence of 208.8 cases per 100,000 population, reflecting a 14.1% increase since 1990 (GBD 2021 Rheumatoid Arthritis Collaborators, 2023). Current evidence suggests RA may be linked to an increased risk of stroke (Nadareishvili et al., 2008). However, the role of RA in stroke is still controversial. A previous meta-analysis showed that RA are associated with increased cardiovascular risk, likely due to the limited number of studies included (only four case-control studies). Simultaneously, they did not analyze the influence of age, sex, country, stroke type, and study design (Lévy et al., 2008). Therefore, this systematic review and meta-analysis aim to evaluate the influence of RA on stroke risk globally.
Methods
This meta-analysis was conducted in accordance with the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines (Page et al., 2021). The protocol was preregistered on the International Prospective Register of Systematic Reviews (PROSPERO) platform (registration number: CRD42024621518).
Data sources and searches
PubMed, Cochrane, and Embase were searched for observational studies published from database inception through October 7, 2025, with no language restrictions. The search strategy combined medical subject headings (MeSH) and keywords, including “Stroke”, “Cerebrovascular Apoplexy”, “Brain Vascular Accident”, “Cerebrovascular Accident”, and “Rheumatoid Arthritis”. The full PubMed search strategy is presented in Table S1 We also reviewed the reference lists of included studies and relevant meta-analyses to identify additional trials.
Eligibility driteria
We included cohort or cross-sectional studies that examined the association between RA and stroke risk, with clear diagnoses of both conditions.
Studies were excluded if they did not report odds ratios (OR) with corresponding 95% confidence intervals (CI). In cases where multiple studies reported data from the same cohort, we selected the study with the longest follow-up or largest sample size. Conference abstracts, study protocols, and duplicate publications were also excluded.
Study selection
Two reviewers (YLL and LXY) independently screened studies for eligibility based on the inclusion and exclusion criteria. Initially, duplicate and irrelevant articles were removed based on titles and abstracts. Full texts of potentially eligible studies were then reviewed to confirm eligibility. Disagreements were resolved through discussion with a third reviewer (WHM).
Data extraction
Data extraction was performed independently by the two primary reviewers (YLL and LXY),following established guidelines for systematic reviews and meta-analyses (Taylor, Mahtani & Aronson, 2021). Data extracted included: first author, publication year, study type, sample size, follow-up duration, participant age and sex, RA and stroke diagnoses, and adjusted confounders. Any discrepancies were resolved by consensus with (WHM).
Risk of bias assessment
Study quality was assessed using the Newcastle-Ottawa Scale (NOS) for cohort studies (Wells et al., 2000), with ratings ranging from 0 to 9 stars. Four stars were allocated for participant selection and exposure measurement, two for comparability, and three for outcome assessment and follow-up adequacy. Studies were classified as low (0–3 stars), moderate (4–6 stars), or high (7–9 stars) quality.
For cross-sectional studies, the American Agency for Health Care Quality and Research (AHRQ) (Rostom et al., 2004) quality evaluation criteria were applied. Each item was scored as ‘1’ for a ‘YES’ response, or ‘0’ for ‘UNCLEAR’ or ‘NO’. The total scores were grouped into low (0–3), moderate (4–7), and high (8–11) quality.
Statistical analysis
The adjusted OR and 95% CI from each study were used to estimate the association between RA and stroke risk. Heterogeneity was assessed using the chi-square test and I2 statistics. Due to differences in the population characteristics and RA duration, there was significant clinical heterogeneity in the included studies. So, regardless of statistical heterogeneity, we would use a random effects model to analyze the data. Sensitivity analyses were conducted by sequentially excluding each study to test the robustness of the results. Publication bias was evaluated visually using funnel plots and statistically with Egger’s test and Begg’s test. We performed subgroup analyses by gender and study type. All statistical analyses were performed using Stata version 17.0 (StataCorp, College Station, TX, USA).
Results
Literature search
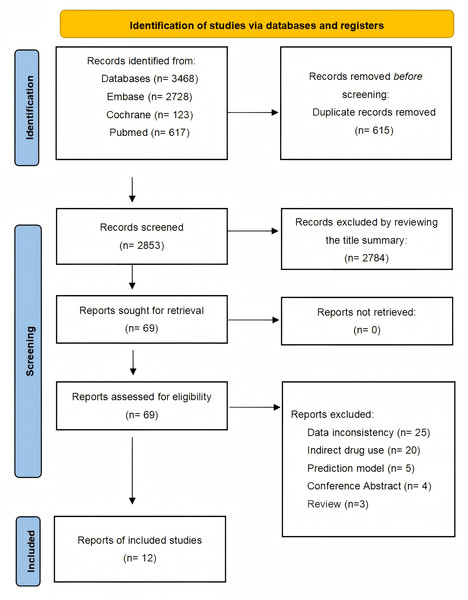
A systematic search of observational studies published before October 7, 2025, identified 3,468 results. After screening titles and abstracts, 15 articles were considered potentially relevant, and 12 studies (Baviera et al., 2021; Chen et al., 2018a; Chen et al., 2018b; Wolfe, Breundlich & Straus, 2003; Kang et al., 2022; Lee et al., 2021; Liou et al., 2014; Logstrup et al., 2021; Shin et al., 2025; Tiosano et al., 2017; Trommer et al., 2021; Zou et al., 2017) were ultimately included. The selection process is detailed in Fig. 1.
Study characteristics
This meta-analysis includes 12 studies (Baviera et al., 2021; Chen et al., 2018a; Chen et al., 2018b; Wolfe, Breundlich & Straus, 2003; Kang et al., 2022; Lee et al., 2021; Liou et al., 2014; Logstrup et al., 2021; Shin et al., 2025; Tiosano et al., 2017; Trommer et al., 2021; Zou et al., 2017) with a total sample size of 1,715,001 individuals, ranging from 2,044 to 818,814 participants. These studies, published between 2003 and 2025, comprised 10 cohort studies (Baviera et al., 2021; Chen et al., 2018a; Chen et al., 2018b; Wolfe, Breundlich & Straus, 2003; Kang et al., 2022; Lee et al., 2021; Liou et al., 2014; Logstrup et al., 2021; Shin et al., 2025; Trommer et al., 2021) and 2 cross-sectional studies (Tiosano et al., 2017; Zou et al., 2017). Eight studies were conducted in developed countries (USA, Germany, Italy, Denmark, Israel, Korea) and four in developing countries (China, Taiwan). Age, sex, and comorbidities were the most common potential confounders in the adjusted models. Key study characteristics are presented in Table 1.
Figure 1: Studies screening process.
| Author | Year | Country | Study type | Sample size | Follow-up years | Age (Mean ± SD) | Sex | Diagnosis of RA/stroke | Confounders adjusted | NOS scores |
|---|---|---|---|---|---|---|---|---|---|---|
| Wolfe, Breundlich & Straus (2003) | 2003 | USA | Retrospective cohort | Total: 11,572 | 6 months | 59.8 ± 13.0 66.0 ± 11.22 | Male 9,093 (23.1%) RA Male 2,479 (18.3%) OA | ICD-10 | Age, sex, education level, smoking, income, hypertension, and body mass index. | 8 |
| Lee et al. (2021) | 2021 | Korea | Retrospective cohort | Total: 16,590 Stroke: 64 | 12 averages | 54 ± 8.7 | Male 735 (26.6%) RA Male 3,675 (26.6%) Control | ICD-10 | Age, sex, and comorbidities, including hypertension, diabetes mellitus, and dyslipidemia. | 9 |
| Kang et al. (2022) | 2022 | Korea | Retrospective cohort | Total: 818,814 Stroke: 1,830 | 8 averages | 54.6 ± 11.6 | Male 36,075 (26.4%) RA Male 180,375 (26.4%) Controls Female 100,394 (73.6%) RA Female 501,970 (73.6%) Controls | ICD-10-CM | Age, sex, smoking, alcohol drinking, regular exercise, obesity, DM, hyperlipidemia and income. | 7 |
| Trommer et al. (2021) | 2021 | Germany | Retrospective cohort | Total: 58,212 Stroke: 842 | 16 averages | 54.8 ± 14.4 | Male 29,106 (35.0%) Female 29,106 (65.0%) | ICD-10 | Age, sex. | 8 |
| Logstrup et al. (2021) | 2020 | Denmark | Retrospective cohort | Total: 90,192 | 22 averages | NA | Female 10,037 (66.8%) RA Female 50,185 (66.8%) Controls | ICD-10 | Age, sex and co-morbidity. | 8 |
| Baviera et al. (2021) | 2021 | Italy | Retrospective cohort | Total: 516,047 Stroke: 10,476 | 13 averages | 59 ± 14.3 RA 45.3 ± 19.1 No RA | Female 11,690 (72.8%) RA Female 255,803 (51.1%) Non-RA | ICD-9-CM/ ICD-10 | Age, sex, index year and comorbidities. | 8 |
| Chen et al. (2018a) | 2018 | Taiwan | Retrospective cohort | Total: 52,840 Stroke: 114 | 6 averages | NA | Male 2,742 (25.95%) RA Male 10,968 (25.95%) Non-RA | ICD-9-CM | Age, sex, hypertension, diabetes mellitus, hyperlipidemia, cancer, mild liver disease, COPD, glucocorticoids, and biologics. | 7 |
| Zou et al. (2017) | 2017 | China | Cross-sectional study | Total: 2,044 Stroke: 56 | 11 averages | 58.5 ± 0.4 Controls 57.8 ± 0.4 RA | Female 702 (68.7%) | ICD-10 | NA | 7 |
| Tiosano et al. (2017) | 2017 | Israel | Cross-sectional study | Total: 69,755 Stroke: 4,361 | NA | ≤65y: 49.8 ± 13.2 >65y: 75.8 ± 7.09 | ≤65y: Female 5,115 (76.7%)RA 25,454 (76.7%) No RA >65y: Female 3,988 (77.9%) RA 19,135 (77.3%) No RA | ICD-10 | NA | 8 |
| Liou et al. (2014) | 2014 | Taiwan | Retrospective cohort | Total: 30,570 Stroke: 383 | 4 averages | NA | Male 1,752 (28.7%) RA 7,008 (28.7%) Control Female 4,362 (71.3%) RA 17448 (71.3%) Control | ICD-9-CM | Age, gender, urbanization level, hyperlipidemia, coronary heart diseases, congestive heart failure, renal disease, atrial fibrillation, valvular heart disease. | 7 |
| Chen et al. (2018b) | 2018 | Taiwan | Retrospective cohort | Total: 3,190 Stroke: 521 | 6 averages | 71.42 ± 10.93 RA 71.34 ± 10.96 No RA | Male 300 (47.02%) RA 1,200 (47.02%) Control | ICD-9-CM | Age, sex, urbanicity, NIHSS, chronic kidney disease. | 8 |
| Shin et al. (2025) | 2025 | Korea | Retrospective cohort | Total: 45,175 Stroke: 3950 | 5 averages | 57.4 ± 9.6 | Male 43,956 (24.3%) | ICD-10 | Sex, age, income, smoking, alcohol drinking, physical activity, and obesity, hyperlipidemia, chronic kidney disease, and atrial fibrillation. | 8 |
Quality assessment
We assessed the quality of all 12 studies using the NOS and AHRQ scales, with scores summarized in Table 1. The average NOS score for cohort studies was 7.75, with each cohort study scoring 7 or higher, indicating high quality. Cross-sectional studies received a score of 8, reflecting high quality according to the AHRQ criteria.
Rheumatoid arthritis and risk of stroke
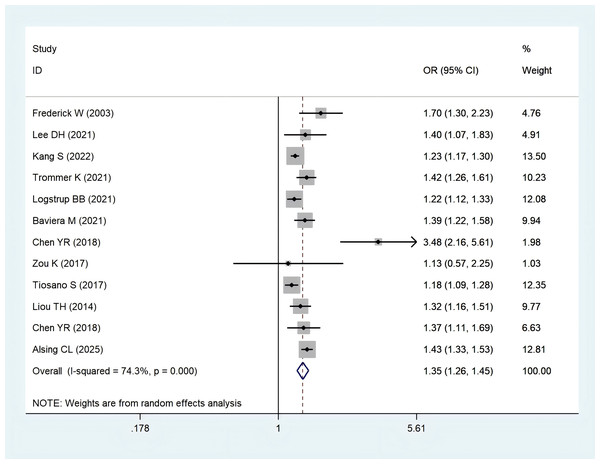
Twelve studies (Baviera et al., 2021; Chen et al., 2018a; Chen et al., 2018b; Wolfe, Breundlich & Straus, 2003; Kang et al., 2022; Lee et al., 2021; Liou et al., 2014; Logstrup et al., 2021; Shin et al., 2025; Tiosano et al., 2017; Trommer et al., 2021; Zou et al., 2017) examined the relationship between rheumatoid arthritis (RA) and stroke risk. The pooled odds ratio (OR) was 1.35 (95% CI [1.26–1.45]; P = 0.000) (Fig. 2). Sensitivity analysis confirmed the robustness of these results, as none of the individual studies reversed the pooled effect size (Fig. S1).
Figure 2: Meta-analysis of the risk of stroke caused by RA.
Studies: Baviera et al. (2021); Chen et al. (2018a); Chen et al. (2018b); Wolfe, Breundlich & Straus (2003); Kang et al. (2022); Lee et al. (2021); Liou et al. (2014); Logstrup et al. (2021); Shin et al. (2025); Tiosano et al. (2017); Trommer et al. (2021); Zou et al. (2017).| Subgroups | Included studies | OR (95% CI) | Heterogeneity | P value of pooled effect | |
|---|---|---|---|---|---|
| I2 (%) | P-values | ||||
| Sex | |||||
| Female | 5 | 1.60 (1.19, 2.16) | 92.8% | 0.000 | 0.002 |
| Male | 5 | 1.44 (1.08, 1.94) | 85.6% | 0.000 | 0.015 |
| Study type | |||||
| Retrospective cohort | 10 | 1.38 (1.28, 1.50) | 75.3% | 0.000 | 0.000 |
| Cross-sectional | 2 | 1.18 (1.09, 1.28) | 0.0% | 0.902 | 0.000 |
| Age | |||||
| ≤65 | 2 | 1.56 (1.21, 2.03) | 53.8% | 0.141 | 0.001 |
| >65 | 3 | 1 .24 (1.02, 1.50) | 73.7% | 0.022 | 0.032 |
| Stroke type | |||||
| Ischemic stroke | 3 | 1.75 (1.27, 2.39) | 84.0% | 0.002 | 0.001 |
| Hemorrhagic stroke | 3 | 1.30 (1.14, 1.48) | 0.0% | 0.407 | 0.000 |
| Country | |||||
| Developed country | 8 | 1.32 (1.23,1.42) | 72.7% | 0.001 | 0.000 |
| Developing country | 4 | 1.59 (1.15,2.20) | 72.7% | 0.002 | 0.005 |
Subgroup analysis
Subgroup analyses by age, sex, and study design showed no significant difference in the association between RA and stroke risk based on study type. However, women with RA appeared to have a slightly higher stroke risk compared to men.
The pooled adjusted odds ratio for ischemic stroke in RA patients was 1.75 (95% CI [1.27–2.39]; P = 0.001). In patients aged over 65 years, the stroke risk was estimated at 1.24 (95% CI [1.02–1.50]; P = 0.032). (Table 2).
Publications bias
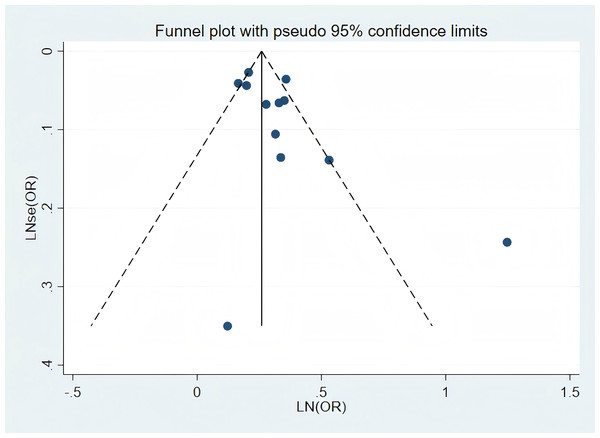
Visual inspection of the funnel plot revealed no significant publication bias in the association between RA and stroke risk (Fig. 3). Similarly, Publication bias was formally assessed using Begg’ s test (P = 0.244; Fig. S2) and Egger’s test (P = 0.067; Fig. S3), which was corrected using the trim-and-fill method (Fig. S4).
Figure 3: Publication bias of the risk of stroke caused by RA.
Discussion
Main findings
This meta-analysis included 12 studies encompassing a total of 1,715,001 individuals, providing a comprehensive evaluation of the association between RA and stroke. We observed a significant increase in stroke risk among individuals with RA, with a 1.35-fold higher risk compared to non-RA controls.
Interpretation of findings
The prevalence of both RA and stroke continues to rise, with cardiovascular disease being a leading cause of mortality in individuals with RA. Therefore, our study aimed to further investigate this association, offering more robust evidence based on a larger number of cohort and cross-sectional studies. Additionally, we conducted a subgroup analysis to assess the impact of RA on different types of stroke, which contributes to a deeper understanding of their specific effects. Our findings emphasize the importance of recognizing RA as a significant risk factor for stroke, supporting the need for tailored healthcare strategies for individuals with RA.
An alternative investigation examined the study on cardiovascular risk factors in a population with rheumatoid arthritis (McEntegart et al., 2001; Semb et al., 2010). However, the lack of an identified association between RA and stroke risk in their analysis was likely due to an underpowered study. In contrast, our study reanalyzed the relationship between RA and stroke, revealing a clear increase in stroke risk in this population.
The precise mechanism linking RA and stroke remains under ongoing investigation. RA is a systemic autoimmune disorder characterized by chronic inflammation of the smaller synovial joints, which causes pain and deformities (Behrouz, 2014). Chronic inflammation in RA accelerates atherosclerosis via endothelial dysfunction, infiltration of inflammatory cells in plaques, and acute plaque rupture, ultimately contributing to stroke risk (Hansson, 2005; Sattar et al., 2003). Moreover, RA can induce necrotizing vasculitis in small- and medium-sized vessels, particularly affecting cerebral vessels (Makol, Matteson & Warrington, 2015). Small-vessel vasculitis is commonly diagnosed through clinical suspicion, MRI evidence of parenchymal or meningeal pathology, and inflammation markers in the cerebrospinal fluid (Makol, Matteson & Warrington, 2015).
RA also has important cardiovascular implications. Several studies have shown an increased prevalence of atrial fibrillation (AF) in RA patients (Engelmann & Svendsen, 2005; Guo, Lip & Apostolakis, 2012; Ungprasert, Srivali & Kittanamongkolchai, 2017), with multifactorial mechanisms that increase the risk of clot formation and, consequently, stroke (Lindhardsen et al., 2012). Furthermore, chronic inflammation in RA patients raises the risk of valvular nodules and thickening, which are associated with a higher risk of stroke (Corrao et al., 2013).
In autoimmune diseases like RA, the occurrence of cardiovascular events is influenced by both traditional cardiovascular risk factors, such as smoking, dyslipidemia, diabetes, and hypertension, as well as by the disease’s systemic effects (Anyfanti et al., 2024). Additionally, some studies suggest that genetic susceptibility may further elevate cardiovascular risk in RA patients (Farragher et al., 2008; Van den Oever, Van Sijl & Nurmohamed, 2013; Yuan et al., 2022).
Although the association between RA and stroke is well-established, the precise mechanisms underlying this relationship warrant further investigation. A more comprehensive understanding of these mechanisms is crucial for mitigating stroke risk and improving outcomes for RA patients who experience stroke.
Implications and limitations
Our meta-analysis synthesizes existing evidence regarding the link between RA and stroke risk. It underscores the importance of closely monitoring stroke risk in RA patients, which may lead to early identification of high-risk individuals and more proactive interventions. One of the strengths of our study is that it is the first systematic review to consolidate the current best evidence on this association. However, there are certain limitations. First, the majority of the included studies were retrospective, which can introduce inherent biases. Additionally, our meta-analysis did not include covariate analysis, and the adjustment factors used in the studies varied. Some studies did not apply any adjustments, which could influence the precision of the results. Despite these variations, most studies included in our review accounted for confounding factors, thereby reducing bias and ensuring the reliability of our findings. This strengthens the clinical applicability of our conclusions.
Conclusions
This meta-analysis indicates that RA is associated with an increased risk of stroke. However, a deeper understanding of the underlying pathophysiology of this association is necessary.