Genotypic analyses and virulence characterization of Glaesserella parasuis isolates from Taiwan
- Published
- Accepted
- Received
- Academic Editor
- Elliot Lefkowitz
- Subject Areas
- Microbiology, Veterinary Medicine
- Keywords
- Glaesserella parasuis, Virulence-associated trimeric autotransporter (vtaA), Genotyping, Enterobacterial repetitive intergenic consensus polymerase chain reaction (ERIC-PCR), Haemophilus parasuis
- Copyright
- © 2019 Lin et al.
- Licence
- This is an open access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, reproduction and adaptation in any medium and for any purpose provided that it is properly attributed. For attribution, the original author(s), title, publication source (PeerJ) and either DOI or URL of the article must be cited.
- Cite this article
- 2019. Genotypic analyses and virulence characterization of Glaesserella parasuis isolates from Taiwan. PeerJ 7:e6960 https://doi.org/10.7717/peerj.6960
Abstract
Background
Glaesserella (Haemophilus) parasuis (G. parasuis) causes severe economic losses in the swine industry. Multiple G. parasuis strains can exist in single animals. Typing techniques are required for identifying G. parasuis isolates. Different strains within a serovar display varying virulence. Enterobacterial repetitive intergenic consensus polymerase chain reaction (ERIC-PCR) can assess the heterogeneity. The group 1 virulence-associated trimeric autotransporters (vtaA) gene is an indicator of virulence. The aim of this study was to characterize Taiwanese G. parasuis isolates via molecular serotyping, vtaA PCR and ERIC-PCR.
Methods
One hundred and forty-five strains were collected between November 2013 and March 2017 in Taiwan and further examined by molecular serotyping, vtaA PCR and ERIC-PCR.
Results
The dendrogram revealed heterogeneous genetic diversity within many clusters. Partial correlation between the ERIC-PCR clusters of different strains, serovars and lesion patterns was observed. Twelve herds (8.3%) infected with more than one strain. Group 1 vtaA positive rate reached 98.6%.
Discussion
This study showed the high genetic diversity of G. parasuis in Taiwan by a high discriminatory capability of ERIC-PCR. Group 1 vtaA commonly exists in G. parasuis isolates and may play important roles in the pathogenesis of Taiwanese G. parasuis isolates.
Introduction
Haemophilus parasuis (H. parasuis) is the causative agent of Glässer’s disease, which induces sudden death, polyserositis, polyarthritis, meningitis, pneumonia and poor production performance, causing severe economic losses in the swine industry (Zhang et al., 2014). H. parasuis has been recently renamed in the NCBI tazonomy as Glaesserella parasuis (G. parasuis) under the family Pasteurellaceae (Inzana, Dickerman & Bandara, 2016). G. parasuis is commonly found in the porcine upper respiratory tract as part of the resident microbiota (Moller & Kilian, 1990). Previous studies have shown that multiple strains can be isolated from individual pig farms, even from single animals (Lin et al., 2018; Turni & Blackall, 2010). Therefore, accurate and effective typing techniques are required for identifying G. parasuis isolates. The isolates of G. parasuis have been classified into 15 serovars via classical immunological methods, including gel immuno-diffusion assays and indirect hemagglutination assays, but approximately 15–40% of pathogenic isolates are non-typable (NT) isolates, with NT rates varying by country or region (Del Rio, Gutierrez & Rodriguez Ferri, 2003; Kielstein & Rapp-Gabrielson, 1992). A molecular serotyping assay based on genetic differences among the capsule loci of the 15 serovar reference strains was developed to differentiate 14 of the 15 serovars of G. parasuis including NT isolates (Howell et al., 2015). Nevertheless, a variety of G. parasuis isolates belonging to the same serovar exhibit varying levels of virulence (Aragon et al., 2010; Olvera, Segales & Aragon, 2007). Non-typable isolates were also found in Taiwan using molecular serotyping (Lin et al., 2018). Although serotyping is useful for vaccine development and vaccine-based strategies, this method is not discriminative enough for differentiating between G. parasuis isolates.
One of the genotyping methods developed to assess the heterogeneity of G. parasuis isolates is enterobacterial repetitive intergenic consensus polymerase chain reaction (ERIC-PCR), which determines the relationships of G. parasuis isolates by comparing genetic fingerprints (Oliveira, Blackall & Pijoan, 2003; Rafiee et al., 2000). Compared to other genotyping assays, such as restriction fragment length polymorphism, multilocus sequence typing and single-locus sequence typing, ERIC-PCR has higher discriminatory power and lower cost (Jablonski et al., 2011; Olvera, Segales & Aragon, 2007).
Virulence-associated trimeric autotransporters (vtaA), belonging to the type Vc secretion system, which are divided into three groups may contribute to the adhesive/invasive capacity of G. parasuis (Olvera et al., 2010). Group 3 vtaA genes are highly conserved among virulent and non-virulent G. parasuis isolates, and group 1 vtaA genes are a potential indicator of virulent isolates (Olvera et al., 2012), making these useful targets in this present study. G. parasuis is a common pathogen that causes economic losses but no published research has been performed to assess the genetic lineages of G. parasuis isolates in Taiwan. Therefore, the aim of this present study was to characterize Taiwanese G. parasuis isolates via molecular serotyping, vtaA PCR, and ERIC-PCR.
Materials and Methods
Vertebrate animal study
The study did not involve any animal experiment. This paper is an extension of a previous study (Lin et al., 2018). The Institutional Animal Care and Use Committee (IACUC) of National Pingtung University of Science and Technology did not deem it necessary for this research group to obtain formal approval to conduct this study.
Bacterial isolates and culture conditions
A total of 145 G. parasuis strains were collected from 302 lesions of 166 diseased pigs. The animals were distributed across 130 diseased-pig herds on 97 pig farms (File S1); samples were collected between November 2013 and March 2017 in Taiwan. Lesions suspected of being caused by G. parasuis, including lesions in the meninges, pleura, pericardia, peritonea, synovial cavities and lungs, were examined by culturing on chocolate agar (at 37 °C, in 5% CO2, and for 18–72 h depending on growth-rate variation among the isolates).
The bacterial isolates were identified by colony morphology, Gram staining (gram-negative bacillus), vtaA group 3 and molecular serotyping species-specific PCR results (Howell et al., 2015; Pina et al., 2009). G. parasuis isolates were stored at −80 °C. A loopful of bacteria from a passaged plate of pure culture was resuspended in 30 μL of ultrapure H2O, which was heated to 100 °C for 30 min and centrifuged at 4,000×g for 1 min. The supernatant was used in the PCR.
Pathological examination
Sick animals or fresh, complete carcasses were subjected to necropsy for gross morphological examination and H&E staining. Histopathological examination focused primarily on meningeal, pleural, pericardial, peritoneal and synovial cavities of joints and lungs. Pathological lesion patterns were determined to respiratory lesion, serositis of Glässer’s disease and both lesions as in a previous study (Lin et al., 2018).
ERIC-PCR assays
Enterobacterial repetitive intergenic consensus polymerase chain reaction was performed using an ABI 2720 (Applied Biosystems, Carlsbad, CA, USA) and a protocol that was modified from a previously published method (Dijkman et al., 2012). Each PCR was performed in a total volume of 30 μL containing ultrapure H2O, 1× DreamTaq buffer, 250 μM dNTP, 0.17 μM primers ERIC-1F (5′-ATGTAAGCTCCTGGGGATTCAC-3′) and ERIC-2R (5′-ATGTAAGCTCCTGGGGATTCAC-3′), 1.25 U of DreamTaq DNA polymerase (Thermo Fisher Scientific, Waltham, MA, USA) and one μL of supernatant. The thermocycling conditions were as follows: 5 min at 94 °C; 40 cycles of 30 sec at 94 °C, 1 min at 50 °C and 2.5 min at 72 °C; and a final extension at 72 °C for 7 min.
The PCR products were stained with ethidium bromide and analyzed using a 20-cm-long 2% agarose gel. For electrophoresis, the gel was subjected to an electric field of six V/cm (300 V, 50-cm full-length electric field) for 3 h. A 50-bp DNA ladder RTU (GeneDireX) and Bio-1D++ software (Vilber Lourmat, Collégien, France) were used to estimate molecular size.
Molecular serotyping
Molecular serotyping assay for G. parasuis isolates was modified from a previously published method using the ABI 2720 (Applied Biosystems, Carlsbad, CA, USA) (Howell et al., 2015). An sp-sp amplicon was used as an internal control. Serotyping results of 279 isolates were from previous study (Lin et al., 2018).
Virulence-associated trimeric autotransporters multiplex PCR (vtaA mPCR) assays
The mPCR protocol for group 1 and group 3 vtaA genes was modified from a previously published method using an ABI 2720 (Applied Biosystems, Carlsbad, CA, USA) (Olvera et al., 2012). Group 3 vtaA is highly conserved among invasive and non-invasive isolates, so this gene was employed for G. parasuis identification. Group 1 vtaA has been mainly detected only in virulent isolates (Pina et al., 2009). Polymerase chain reaction was performed in a total volume of 30 μL containing ultrapure H2O; 1× DreamTaq buffer; 250 μM dNTP; 0.17 μM YADAF1 (5′-TTTAGGTAAAGATAAGCAAGGAAATCC-3′), PADHR1 (5′-CCACACAAAACCTACCCCTCCTCC-3′), YADAF3 (5′-AATGGTAGCCAGTTGTATAATGTTGC-3′) and PADHR3 (5′-CCACTGTAATGCAATACCTGCACC-3′) primers; 1.25 U of DreamTaq DNA polymerase (Thermo Fisher Scientific, Waltham, MA, USA) and one μL of supernatant. The thermocycling conditions were as follows: 5 min at 94 °C; 35 cycles of 30 sat 94 °C, 30 s at 60 °C and 1 min at 72 °C; and a final extension at 72 °C for 7 min. The PCR products were stained with ethidium bromide and analyzed using a 2% agarose gel.
Data analysis
The G. parasuis fingerprints were compared and analyzed using the unweighted pair group method with arithmetic mean with a 5% homology coefficient by using Bio-1D++ software (Vilber Lourmat, Collégien, France) to define strains. The cluster analysis was performed at a genetic similarity of 80%.
Results
Genotyping of G. parasuis by ERIC-PCR
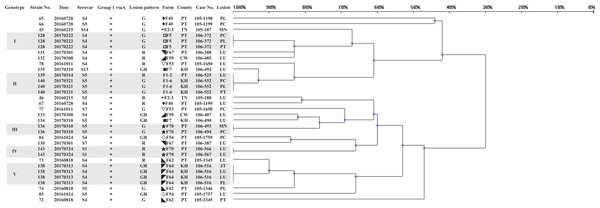
Three hundred and two isolates were identified as 145 strains, named strain 1–145, by same patterns of ERIC-PCR from same infected herds. Eighteen strains (12.4%) could not be typed using molecular serotyping due to absence of any serovar specific amplicon (abbreviated as NT). The results of the correlated clusters are depicted in Fig. 1. The ERIC-PCR dendrogram revealed a high level of genetic heterogeneity in many clusters. Two major clusters including 118 strains (81.4%) were observed in the dendrogram. Only 10 strains (6.9%) were clustered in five identical genotypes.
Figure 1: Dendrogram showing relationships of and information on 145 G. parasuis strains.
Clinicopathological outcomes were divided into three types, namely, serositis of Glässer’s disease (G), lower respiratory infection (R) and serositis with respiratory lesions (GR). County name abbreviations: Changhua (CW), Yunlin (YL), Tainan (TN), Kaohsiung (KH), Pingtung (PT), Hualien (HL). Lesions represent the origins of the strains. Lesion abbreviations: meninges (MN), pleura (PL), pericardium (PC), peritoneum (PT), joint (JT) and lung (LU). Different symbols represent strains from the same pig farm.Upon applying an 80% similarity, 37 clusters were obtained. Partial correlation between the ERIC-PCR clusters of different strains, serovars and lesion patterns was observed. Some clusters included strains from one major serovar (proportion is greater than 70%), including clusters 1, 2, 3, 6, 15, 17, 18, 25, 26, 32 and 35. Some clusters included strains from one main lesion pattern (proportion is greater than 70%), including clusters 1, 3, 5, 6, 7, 9, 13, 15, 16, 18, 25, 28, 31, 33 and 35.
Strains 139 and 140, typed as an identical genotype and serovar 5, were isolated on similar dates and belonged to a same swine production system. Nevertheless, the lesion patterns of strains 139 and 140 were different. In farm F1-2, this strain was found to cause only respiratory lesions; however, this strain was found to induce polyserositis in farm F1-6 after one week. Although the nursery farms F1-2 and F1-6 are located in different counties, the weaning pigs were from the same sow farm. The spread of G. parasuis strains could be tracked according to the genotype.
The strains causing diseases on same farms at different time periods belonged to various clusters, like farms F5 (◘) and F75 (☆). In farm F75, there were a total of five different strains belonging to different clusters in one year. Of total 11 strains in 5 years from farm F5, 2 and 3 strains belonged to clusters 20 and 24, respectively, but they were still different strains, even different serovars (Table 1).
| Farm | Strain no. | Cluster | Serovar |
|---|---|---|---|
| F5 | 1 | 24 | 4 |
| 3 | 24 | 4 | |
| 8 | 24 | 4 | |
| 28 | None | 13 | |
| 37 | 20 | 4 | |
| 71 | 20 | NT | |
| 79 | None | 5 | |
| 90 | None | 4 | |
| 97 | 12 | NT | |
| 111 | 35 | 13 | |
| 128 | 17 | 4 | |
| F75 | 54 | 28 | 5 |
| 89 | 31 | 5 | |
| 99 | 3 | 4 | |
| 110 | 33 | NT | |
| 142 | 22 | 9 |
From 12 herds (8.3%), there were 50 isolates (16.6%), identified as 27 strains (18.6%), which were isolated as more than one strain from same period endemic herds or same individuals. Some isolates were identified as different strains by both molecular serotyping and ERIC-PCR. Others isolates belonging to same serovars were identified as different strains via ERIC-PCR, namely, strains 65, 67, 72, 73, 74, 132 and 133. The others belonging to NT, strains 102, 103, were also differentiated by ERIC-PCR.
A panel of 33 isolates, identified as 24 strains by ERIC-PCR, was employed as a template to illustrate the relationship among isolates, strains, serovars, farms and infected organs (Fig. 2). Most isolates from same animal or farm of origin were clustered in identical genotypes and typed within same serovar, namely, strains 128, 136, 138, 140 and 143 (data for other isolates were similar), and had homologous patterns (Figs. 2 and 3). The relationship between strains 139 and 140 was mentioned above. The other isolates from same period endemic herds or same individuals, as shown in Fig. 2, were determined to be heterogeneous via genotyping with or without serotyping.
Figure 2: Dendrogram showing relationships of and information on 33 G. parasuis isolates identified as 24 strains.
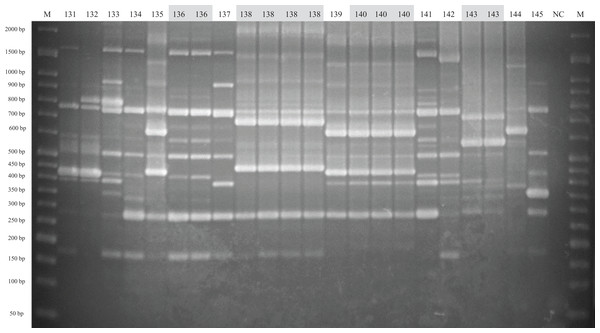
The method of analysis and the meaning of the symbols are the same as in Fig. 1.Figure 3: ERIC-PCR gel image of G. parasuis isolates belonging to strains 131 to 145.
Lane M: 50-bp DNA Ladder RTU (GeneDireX); Lane NC: negative control.Virulence-associated trimeric autotransporters multiplex PCR
The group 1 and group 3 vtaA genes were detected via vtaA mPCR. A total of 145 strains were positive for group 3 vtaA (100%) and the molecular serotyping species-specific amplicon. Only two strains (1.4%) were negative for group 1 vtaA. One strain belonging to serovar 13 was isolated from the synovial cavity of an animal with pleuritis and arthritis. The other strain typed as serovar 5 was isolated from the pericardium of an animal with pleuritis and epicarditis. The positive rate of group 1 vtaA was 98.6%.
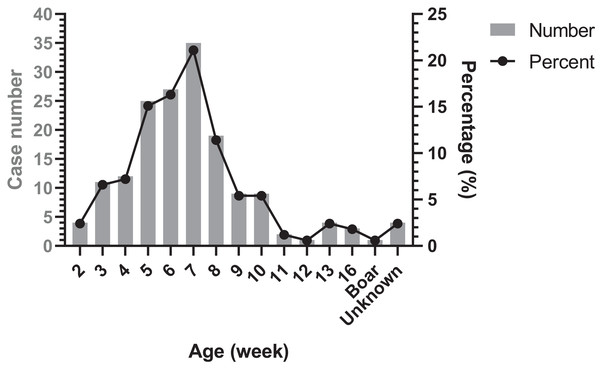
Occurrence period of Glässer’s disease
The age distribution of Glässer’s disease was shown in Fig. 4. The highest occurrence period was from 5 to 8 week-old in nursery, followed by suckling period. However, there were still Glässer’s disease cases in growing pigs and even in breeding period.
Figure 4: Age distribution of diseased pigs.
Discussion
To the authors’ knowledge, this is the first study to describe the genetic diversity of G. parasuis in Taiwan. The ERIC-PCR dendrogram shows G. parasuis strains are genetically heterogeneous within the same serovar as well as between serovars. The great discriminatory power and typeability of ERIC-PCR are cited by previous studies of G. parasuis (Dijkman et al., 2012; Jablonski et al., 2011; Moreno et al., 2011; Oliveira, Blackall & Pijoan, 2003). In our study, ERIC-PCR is used to differentiate isolates within and between serovars, typed by molecular serotyping mPCR, which still remains molecular serotyping NT G. parasuis isolates in Taiwan (Lin et al., 2018). Therefore, the utility of ERIC-PCR for epidemiological survey is greater than that of not only immunological serotyping assays but also molecular serotyping assays.
In our data of strains from diseased animals, partial relationship is observed between genotypes by ERIC-PCR and serovars by molecular serotyping but the correlation is not obligate. Previous reports have indicated that the serovars are based on differences in the capsule genes of G. parasuis (Howell et al., 2013). Nonetheless, ERIC-PCR patterns can be obtained from the whole bacterial genome (Olvera, Segales & Aragon, 2007). The gap between these two typing assays may be caused by the scope of gene detection. Some strains isolated from different farms and at different times belong to the same serovars typed in the same clusters, which was likely due to homologous whole bacterial genomes. This may be related to the dissemination and evolution of G. parasuis. Additional data, such as data regarding antibiotic resistance and virulence genes, are worth collecting to better elaborate the epidemiology of G. parasuis for disease prediction, investigation, prevention and control in the future.
Cross-protection between strains belonging to the same serovar has been evidenced by several studies (Miniats, Smart & Ewert, 1991; Smart & Miniats, 1989; Takahashi et al., 2001). Nonetheless, heterologous protection across strains and serovars has been reported as being sporadic and inconsistent (Oliveira & Pijoan, 2004). Autogenous bacterins are considered an effective strategy against Glässer’s disease (Smart, Hurnik & Macinnes, 1993). In this case, ERIC-PCR, which has great discriminatory power, can be a useful tool for isolate identification.
In Taiwan, porcine multiple-site production models are more popular. Many nursery farms, which have been modified from farrow-to-finish facilities, are owned by agricultural corporations and obtain weaning pigs from same sow farms. This phenomenon is common in Asian countries of which swine industries are developing. Sows are reservoirs of G. parasuis and transmit G. parasuis to suckling pigs (Nedbalcova et al., 2011). The results show pigs from two nursery pig farms sharing a sow farm are infected with the same G. parasuis strain at the same time. In addition, maternal immunity is an important factor influencing outbreaks of Glässer’s disease (Nedbalcova et al., 2011). Furthermore, some swine production model usually mixes weaning pigs from different origins. Therefore, epidemiological monitoring of multiple-site pig farms and establishing herd immunity against G. parasuis strains is important for swine production systems for the prevention and control of Glässer’s disease. Previous study indicates sows are reservoirs and passive antibodies can prevent Glässer’s disease (Nedbalcova et al., 2011). Vaccination of sows will be an efficacious strategy to control Glässer’s disease in suckling and weaning pigs. However, maternal antibody cannot protect late nursery and growing pigs. Vaccination of piglets can be considered to decrease economic losses caused by death and feed efficiency reduction. Our data showed various genotypes of G. parasuis causing Glässer’s disease on the same farms at different time periods or same period endemic herds. Virulence varying within same serovars is caused by genome differences and existence of virulence genes. The cross-protection among different genotypes belonging to same serovars should be concerned. Distinct clinicopathological outcomes in different farms caused by identical G. parasuis strains may be caused by various factors, such as room temperature, ventilation and coinfection with other pathogens.
It is well established that various strains can be isolated from individual pig farms, even from single animals (Cerda-Cuellar et al., 2010; Oliveira, Blackall & Pijoan, 2003; Olvera, Calsamiglia & Aragon, 2006a; Olvera, Cerda-Cuellar & Aragon, 2006b; Ruiz et al., 2001). ERIC-PCR can be used to identify isolates to assist G. parasuis recovery in challenge trials, sample collection for antibiotic resistance investigations, determination of antibiotic strategies for diseases causing by more than one strain and other studies requiring great discriminatory power. Although ERIC-PCR cannot offer information as valuable as whole-genome sequence data and comparisons of results among different laboratories is problematic, ERIC-PCR is still a useful, inexpensive, fast and practical tool for epidemiological surveillance, and in the identification of the source of transmission as an initial screening genotyping method. In the future, after typing by ERIC-PCR, molecular serotyping NT isolates are worthy of sequence analyses of polysaccharide biosynthesis loci. Based on sequence data and serovars by immunological serotyping assays, the gene functions, cross-protection among different serovars and new molecular serotyping assays can be investigated. In our study, group 1 vtaA are common in G. parasuis isolates from Taiwanese diseased pigs. This result is similar to the results of previous studies (Dijkman et al., 2012; Moreno et al., 2011; Olvera et al., 2012), so group 1 vtaA may also be important in the pathogenesis of Taiwanese G. parasuis isolates and worthy of developing as vaccine candidates. However, G. parasuis without group 1 vtaA still can be isolated from pigs with serositis (Moreno et al., 2011). Except host and environment factors, the pathogenicity of G. parasuis is considered to be related to multiple virulence factors (Costa-Hurtado & Aragon, 2013). Therefore, G. parasuis without group 1 vtaA may have ability to induce disease if other virulence genes exist and/or animals are immunosuppressive.
Conclusions
This study shows the high genetic diversity of G. parasuis isolates in Taiwan. Enterobacterial repetitive intergenic consensus polymerase chain reaction has better discriminatory capability than molecular serotyping assays for the detection of slight differences among different strains and can be applied as an effective tool in the routine surveillance of G. parasuis for prevention and control strategies and further sequence studies. Group 1 vtaA are common in G. parasuis isolates from diseased pigs and may play important roles in the pathogenesis of Taiwanese G. parasuis isolates.