Interaction between Pseudomonas aeruginosa and Aspergillus fumigatus in cystic fibrosis
- Published
- Accepted
- Received
- Academic Editor
- Katrine Whiteson
- Subject Areas
- Microbiology, Infectious Diseases, Internal Medicine, Respiratory Medicine, Science and Medical Education
- Keywords
- Pseudomonas aeruginosa, Infection, Aspergillus fumigatus, Intermicrobial interaction, Cystic fibrosis
- Copyright
- © 2018 Zhao and Yu
- Licence
- This is an open access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, reproduction and adaptation in any medium and for any purpose provided that it is properly attributed. For attribution, the original author(s), title, publication source (PeerJ) and either DOI or URL of the article must be cited.
- Cite this article
- 2018. Interaction between Pseudomonas aeruginosa and Aspergillus fumigatus in cystic fibrosis. PeerJ 6:e5931 https://doi.org/10.7717/peerj.5931
Abstract
Background
Cystic fibrosis (CF) is a disease characterized by chronic airway infection with a high incidence and poor prognosis. Pseudomonas aeruginosa and Aspergillus fumigatus are pathogens commonly found in CF patients. Clinically, these two microorganisms often coexist in the airway of CF patients. Combined infection with P. aeruginosa and A. fumigatus results in worsening lung function and clinical condition.
Methods
In this review, we focus on the mutual inhibition and promotion mechanisms of P. aeruginosa and A. fumigatus in CF patients. We also summarized the mechanisms of the interaction between these pathogenic microorganisms.
Results
P. aeruginosa inhibits A. fumigatus growth through the effects of phenazines, the quorum sensing system, iron competition, bacteriophages, and small colony variants. P. aeruginosa induces A. fumigatus growth through volatile organic compounds and subbacteriostatic concentrations of phenazines. A. fumigatus interferes with P. aeruginosa, affecting its metabolic growth via phenazine metabolic transformation, gliotoxin production, and reduced antibiotic sensitivity.
Discussion
Coexistence of P. aeruginosa and A. fumigatus can lead to both mutual inhibition and promotion. In different stages of CF disease, the interaction between these two pathogenic microorganisms may shift between promotion and inhibition. A discussion of the mechanisms of P. aeruginosa and A. fumigatus interaction can be beneficial for further treatment of CF patients and for improving the prognosis of the disease.
Introduction
Cystic fibrosis (CF) is the most common inherited lung infection disease; it is estimated that more than 70,000 people worldwide suffer from CF (Cystic Fibrosis Foundation, 2017). As CF affects multiple organs, the morbidity and mortality of CF are caused by airway infection and the associated inflammation (Zhao et al., 2012). Mutations in the cystic fibrosis transmembrane conductance regulator (CFTR) gene result in dysfunction or a lack of CFTR protein and impaired mucociliary clearance in CF patients. CF-related lung disease begins early in life, with inflammation, impaired mucociliary clearance, and consequent chronic infection of the airways (Robinson & Bye, 2002).
The pathogens Pseudomonas aeruginosa and Aspergillus fumigatus are common in lung infections. The bacterium P. aeruginosa infects 70–80% of adult patients with CF (Al-Momani et al., 2016; Salsgiver et al., 2016) and A. fumigatus is the most common fungal pathogen isolated from the airways of CF patients. The reported prevalence of A. fumigatus colonization in CF patients is between 16% and 58% (Amin et al., 2010; Becker et al., 1996; Skov et al., 2005; Stevens et al., 2003; Valenza et al., 2008). Moreover, many studies have shown that the sputum of CF patients contains both P. aeruginosa and A. fumigatus. Previous studies have reported the isolation of A. fumigatus in up to 60% of CF patients with P. aeruginosa infection, and P. aeruginosa has been isolated in up to 64.2% of CF patients with A. fumigatus infection (Bakare et al., 2003; Paugam et al., 2010). The results from a systematic review and meta-analysis showed that the pooled co-colonization prevalence of P. aeruginosa and A. fumigatus in patients with CF was 15.8% (95% CI [9.9–21.8]) with variation ranging between 2.3% and 44.8% (Zhao et al., 2018).
Co-colonization by P. aeruginosa and A. fumigatus in CF patients correlates with a worsened condition (Amin et al., 2010; Shoseyov et al., 2006). For example, an Irish registry analysis showed that P. aeruginosa and A. fumigatus co-colonization was associated with reduced FEV1, more frequent hospitalization, greater respiratory exacerbation, and increased use of anti-microbials compared with patients without the co-existence of these pathogens (Reece et al., 2017). Another study reported increased levels of toxic products in supernatants from P. aeruginosa and A. fumigatus co-culture compared with those from P. aeruginosa monoculture. Indeed, the production of cytotoxic elastase by P. aeruginosa increases in the presence of the filamentous fungus A. fumigatus, damaging human lung epithelial cells, decreasing lung function and facilitating disease progression (Smith et al., 2015).
Pseudomonas aeruginosa and A. fumigatus interact in a complex manner in the airways of co-infected CF patients. In this review, we summarize in detail the mechanisms underlying the interaction between P. aeruginosa and A. fumigatus. We review the principles of mutual inhibition and growth promotion of P. aeruginosa and A. fumigatus as well as interaction between the two microorganisms in CF patients at different stages of the disease, emphasizing the impact of such interactions on the conditions of CF patients. In the presence of co-infection, P. aeruginosa and A. fumigatus do not exist in isolation; instead, they affect each other and combat the immune response together to collaboratively affect the development of the disease.
Survey methodology
The EmBase, PubMed, and Web of Science databases were searched (until January 2018) using the following free-text terms: P. aeruginosa, A. fumigatus, and CF.
Inhibitory effect of P. aeruginosa on A. fumigatus
Pseudomonas aeruginosa inhibits A. fumigatus growth by the effect of phenazines, the quorum sensing (QS) system, iron competition, bacteriophages, and small colony variants (SCVs).
Phenazines constitute a large proportion of the numerous molecules secreted by P. aeruginosa during growth and are considered important virulence factors against target organisms, including other bacteria, fungi, and mammalian cells (Gibson, Sood & Hogan, 2009; Lau et al., 2004; Price-Whelan, Dietrich & Newman, 2006; Whiteson et al., 2014). Phenazines are present in CF patient sputum at concentrations ranging from 1 to 100 μg ml−1 (Wilson et al., 1988), and their increasing concentrations can cause a concomitant decline in lung function (Hunter et al., 2012). In CF patients, overproduction of alginate in P. aeruginosa biofilms generates a hypoxic gradient and anaerobic environment that enhances phenazine toxicity (Wang, Kern & Newman, 2010). P. aeruginosa phenazines have an important impact on electron shuttling, redox chemistry, and biofilm development through the toxic superoxide signaling and generation (Pierson & Pierson, 2010; Price-Whelan, Dietrich & Newman, 2006). Phenazines are regarded as endogenous redox-active molecules that promote P. aeruginosa growth and survival under iron-limiting conditions in CF patients and include five secreted molecules: pyocyanin (5-N-methyl-1-hydroxyphenazine, PYO) (Blyth & Forey, 1971; Kerr et al., 1999; Mangan, 1969), 1-hydroxyphenazine (1-HP) (Kerr et al., 1999; Mangan, 1969), phenazine-1-carboxamide (PCN), phenazine-1-carboxylic acid (PCA) (Briard et al., 2015), and dirhamnolipids (diRhls) (Briard et al., 2017).
The QS system comprises a cell density-based intercellular communication system in which signals are transmitted within the same bacterial species and between different species. The QS system regulates a variety of biological characteristics, including the release of virulence factors. The QS system in P. aeruginosa is involved in the regulation of elastase, pyocyanin, proteolytic enzyme, and biofilm formation (Lee & Zhang, 2015). There are three known QS systems in P. aeruginosa, namely, las, rhl, and pqs.
Fe is a very important element for P. aeruginosa and A. fumigatus growth. In fact, the numerous iron acquisition systems underlie the ability of P. aeruginosa to survive in diverse environments, with a strong ability to compete with other organisms for this essential metallonutrient. There are three classes of pyoverdines, which are iron chelators, with similar iron-binding properties and levels of activity. Type II pyoverdine is the main type involved in P. aeruginosa strains associated with CF (De Vos et al., 2001).
Bacteriophages have an important impact on bacterial virulence and phenotypic variation. It has been shown that the formation of SCVs in biofilms can be mediated by the filamentous bacteriophage Pf4 of the P. aeruginosa strain PAO1 (Mooij et al., 2007), and this morphological type is related to parameters of poor lung function in CF patients.
As SCVs represent a CF P. aeruginosa phenotype, analysis of SCVs isolated during chronic P. aeruginosa colonization in CF patients is a worthy endeavor. For example, it has been reported that SCVs isolated from CF patients are resistant to antibiotics and are associated with poor lung function and a poor clinical condition (Evans, 2015; Hogardt & Heesemann, 2010; Häussler et al., 1999).
Inhibitory effect of phenazines on the growth of A. fumigatus
Pseudomonas aeruginosa-secreted phenazines prevent the growth of A. fumigatus. It is thought that the toxic effects of phenazines on prokaryotes and diverse eukaryotic hosts result from their redox activities or inactivation of oxidative stress response proteins (Hassett et al., 1992; Muller, 2002; O’Malley et al., 2003). In target cells, reduced phenazines are oxidized by NAD(P)H and oxygen to generate reactive oxygen species (ROS), specifically O2·− Moreover, generation of reactive nitrogen species (RNS) is induced by overproduction of O2·− (Martínez & Andriantsitohaina, 2009). Nitric oxide (NO·) is produced by mitochondrial processes, and highly toxic peroxynitrite radicals (ONOO−) are generated via reactions between NO· with O2·− radicals (Martínez & Andriantsitohaina, 2009). Overall, mitochondria are the main target of phenazine-produced ROS and RNS, and phenazines have a significant impact on the mitochondrial ultrastructure of A. fumigatus hyphae. All four phenazines (PYO, PCA, PCN, 1-HP) show A. fumigatus growth inhibitory effects by inducing the production of ROS, specifically O2·−, and the RNS ONOO− (pathway ① in Fig. 1) (Briard et al., 2015).
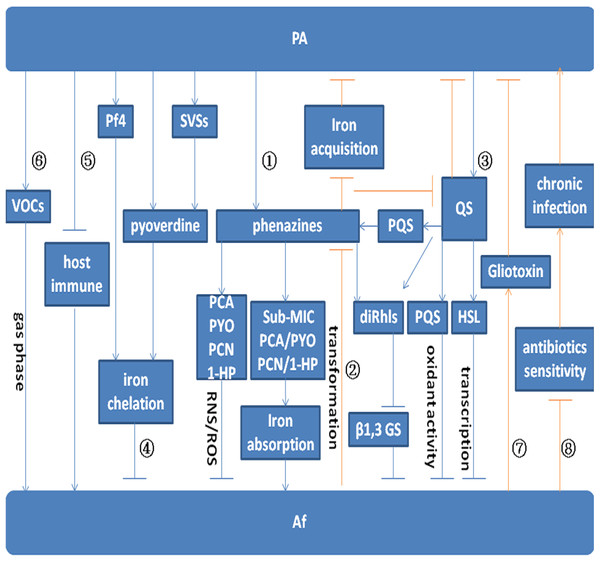
Figure 1: Model for the interaction between P. aeruginosa and A. fumigatus.
Arrows indicate promotion. Arrows without heads indicate inhibition. Blue lines indicate the effect of P. aeruginosa on A. fumigatus. Red lines indicate the effect of A. fumigatus on P. aeruginosa. Pathway ① indicates the effect of P. aeruginosa on A. fumigatus by phenazine. PYO, PCA, PCN, and 1-HP inhibit A. fumigatus growth by inducing the production of ROS and RNS. Sub-MIC PYO, PCA, PCN, and 1-HP promote A. fumigatus growth by iron absorption. A. fumigatus growth can be inhibited by diRhls, which blocks β1,3 GS activity. Pathway ② shows the effect of A. fumigatus on P. aeruginosa by phenazine transformation. The metabolic conversion of phenazine by A. fumigatus inhibits the reduction of Fe3+ and affects QS system regulation in P. aeruginosa. Pathway ③ depicts the inhibition of toxic products and small molecules regulated by the QS system. The QS system in P. aeruginosa inhibits A. fumigatus growth via the effect of diRhls, PQS, and 3-oxo-C12 HSL. Pathway ④ shows that P. aeruginosa inhibits A. fumigatus growth via the effect of pyoverdine, Pf4, and SVSs on A. fumigatus iron deprivation. Pathway ⑤ and Pathway ⑥ illustrate P. aeruginosa promotion of A. fumigatus growth through the inhibition of host immune components and emission of VOCs. Pathway ⑦ shows that gliotoxin produced by A. fumigatus interferes with the metabolic growth of P. aeruginosa. Pathway ⑧ shows that A. fumigatus reduces the sensitivity of P. aeruginosa to antibiotics and promotes chronic infection.Another related study reported that phenazine-derived metabolites acting as interspecies signals can affect filamentous fungal development through oxidative stress regulation (Zheng et al., 2015b). In P. aeruginosa–A. fumigatus co-culture biofilms, development of the latter is differentially modulated by phenazine-derived metabolites of the former. With a decreasing phenazine gradient, A. fumigatus shifts from weak vegetative growth to an asexual sporulation phase (conidiation), and this shift in morphology is correlated with the production of phenazine radicals and concomitant ROS generation by phenazine redox cycling.
DiRhls induce A. fumigatus to produce an extracellular matrix that facilitates P. aeruginosa binding. A. fumigatus growth can be inhibited by diRhls, which blocks the β1,3 glucan synthase (GS) activity (pathway ① in Fig. 1) (Briard et al., 2017).
A recent study reported that a double phenazine mutant was similar to the wild-type organism in terms of its inhibitory power against A. fumigatus (Sass et al., 2018), with little difference caused by the complete lack of phenazine molecules due to mutation. The results of this study are different from those of previous studies. Several previous studies have emphasized the inhibitory power of phenazines on A. fumigatus (Briard et al., 2015, 2017; Zheng et al., 2015b), but a deficiency of these molecules via mutation appeared to cause little difference in this study. This finding suggests that the concentrations of phenazines that have been previously studied in vitro may be irrelevant to those in vivo. In addition, compensation for the loss of phenazine-mediated inhibitory activity by upregulation of other factors in the mutants could not be excluded in this study. Further research is needed to explore the effect of phenazines on the growth of A. fumigatus.
Effects of the inhibition of the QS system on the growth of A. fumigatus
The P. aeruginosa QS network plays a role in inhibiting A. fumigatus growth and biofilm formation (pathway ③ in Fig. 1) (Mowat et al., 2010). The las QS system is essential for the production of the diffusible signaling molecule acyl homoserine lactone (AHL) N-(3-oxododecanoyl)-l-homoserine lactone (3-oxo-C12 HSL) (Smith & Iglewski, 2003), resulting in the expression of specific target genes in P. aeruginosa. In addition, 3-oxo-C12 HSL is one of the AHLs frequently identified in extracts of respiratory secretions from CF patients infected with P. aeruginosa (Smith & Iglewski, 2003). By utilizing two P. aeruginosa QS knockout strains, PAO1:ΔLasI and PAO1:ΔLasR, one study illustrated that 3-oxo-C12 HSL inhibits A. fumigatus biofilm formation (Mowat et al., 2010). The PAO1:ΔLasI strain was unable to synthesize 3-oxo-C12 HSL, whereas PAO1:ΔLasR synthesized 3-oxo-C12 HSL but could not respond to it. Furthermore, A. fumigatus growth was significantly greater when in direct co-culture with PAO1:ΔLasI and PAO1:ΔLasR than in co-culture with wild-type PAO1. The indirect effect of the P. aeruginosa QS knockout strains on A. fumigatus biofilm development was assessed using the Transwell system, which showed significantly less inhibition of A. fumigatus biofilm development when in indirect co-culture with PAO1:ΔLasI and PAO1:ΔLasR than with the wild-type strain. Additionally, the cellular viability of A. fumigatus conidia and the biomass of A. fumigatus biofilms were reduced by diffusible and heat-stable soluble molecules, such as decanol, decanoic acid and dodecanol (structurally similar to the QS molecules produced by P. aeruginosa) in a concentration-dependent manner. At the molecular level, it is likely that these molecules lead to hyphal repression by affecting key transcription factors (Mowat et al., 2010).
In CF patients, P. aeruginosa produces rhamnolipids (Rhls), which are controlled by the QS system. Rhls are largely composed of diRhls and monorhamnolipids, and the diRhls secreted by P. aeruginosa may affect A. fumigatus. For example, diRhls induce A. fumigatus to produce an extracellular matrix that facilitates binding by P. aeruginosa (Briard et al., 2017). As stated above, diRhls also inhibit A. fumigatus growth by blocking β1,3 GS activity and altering cell wall architecture. In the presence of diRhls, A. fumigatus displays multibranched hyphae and a thicker cell wall rich in chitin. This growth phenotype of A. fumigatus is similar to that following treatment with anti-fungal echinocandins. Although the two rhamnose moieties attached to fatty acyl chains are essential structures for the interaction of diRhl with β1,3 GS, the site of β1,3 GS action differs between diRhls and echinocandins. Overall, diRhls and azole anti-fungals exhibit a synergistic anti-fungal effect (Briard et al., 2017).
A recent study reported that alkylhydroxyquinolones (AHQs), autoinducers secreted by P. aeruginosa, could suppress biofilm formation in A. fumigatus. The AHQ interkingdom signaling molecules 2-heptyl-3-hydroxy-4-quinolone (PQS) and 2-heptyl-4-quinolone (HHQ), which are involved in QS in P. aeruginosa, were both able to alter A. fumigatus biofilm biomass and structure (Reen et al., 2016). Both pro- and anti-oxidant activities have been reported for PQS and HHQ. AHQ interkingdom signaling molecules can interact with lipopolysaccharides, cellular membranes, and membrane vesicles in several bacterial species (Häussler & Becker, 2008). Redox-active phenazines of P. aeruginosa, which exhibit inhibitory activity against A. fumigatus growth, are also controlled by AHQs (Moree et al., 2012). Overall, these small interkingdom signaling molecules of P. aeruginosa disrupt A. fumigatus biofilm formation and render A. fumigatus susceptible to clearance by drugs. As these bacterial molecules are selectively non-cytotoxic to host cell lines, they may be used as viable molecular therapeutics.
The anti-A. fumigatus capacity of P. aeruginosa pyoverdine mutants has been assessed in recent studies. Some residual inhibition of A. fumigatus can be detected in pyoverdine mutants, and in addition to the anti-Aspergillus effect of pyoverdine, other inhibitors may contribute to the total fungal inhibition by wild-type P. aeruginosa. Some of these residual inhibitors in pyoverdine mutants may be related to QS-regulated metabolites, such as Rhls. The anti-Aspergillus ability of QS mutants was also examined, with the results showing that QS-regulated metabolites have an important anti-Aspergillus function. Indeed, these metabolites are potential intermicrobial inhibitors. The decreased anti-A. fumigatus activity of QS mutants might be related to loss of the combined activity of many downstream products, and decreases in pyoverdine production in QS mutants may also lead to their reduced anti-Aspergillus capacity (Sass et al., 2018).
Inhibition of A. fumigatus growth by Fe metabolism
Pseudomonas aeruginosa inhibits A. fumigatus growth through Fe limitation, which can, in part, result from the modulation of siderophore production by the fungus due to metabolites from the bacterium (Phelan et al., 2014).
Recent research has shown that P. aeruginosa pyoverdine can suppress A. fumigatus growth and biofilm formation via the chelation of iron, reducing its availability to A. fumigatus (pathway ④ in Fig. 1) (Sass et al., 2018). In this study, pvdD pchE and pvdD mutants (loss of pyoverdine and siderophore), which are defective in inhibiting A. fumigatus growth and biofilm formation in various assays, were evaluated. The inhibitory effect of pyoverdine deletion mutants was restored with pure pyoverdine, and the A. fumigatus sidA mutant that is unable to produce siderophores was found to be hypersusceptible to P. aeruginosa metabolites and to pyoverdine. Thus, the siderophore-deficient A. fumigatus mutant was readily inhibited by P. aeruginosa. Clinical P. aeruginosa isolates derived from the lungs of CF patients have revealed a correlation between the amount of pyoverdine produced and the anti-fungal activity of clinical samples. The results suggest that the siderophore pyoverdine is an important inhibitory molecule (Sass et al., 2018).
As pyoverdine can capture iron from the environment, it can deprive A. fumigatus of the iron that is essential for its growth and metabolism. Iron sequestration by pyoverdine leads to iron starvation and increased siderophore secretion by A. fumigatus (Sass et al., 2018). In a shared microenvironment, P. aeruginosa and A. fumigatus compete for iron to promote their own survival, and high pyoverdine expression antagonizes A. fumigatus metabolism and growth, which might support anti-fungal treatment. Key aspects of the competition between P. aeruginosa and A. fumigatus include the relative amounts of siderophores produced, the speed of siderophore production, and the relative affinity for Fe.
Bacteriophage Pf4 inhibits the metabolic activity of A. fumigatus biofilms
In a recent study, it was reported that the Pf4 phagosome can inhibit A. fumigatus metabolism and growth by binding iron and causing iron deficiency (pathway ④ Fig. 1) (Penner et al., 2016). Pf4 inhibition of A. fumigatus is caused by iron binding and the sequestration of A. fumigatus iron resources (pathway ① Fig. 2), and inhibition of A. fumigatus metabolism by Pf4 can be overcomed with ferric iron supplementation. Moreover, inhibition of A. fumigatus biofilm formation by phages is reversed by low doses of iron, indicating that A. fumigatus is more sensitive to iron inhibition by the Pf4 phage than it is to other elements in P. aeruginosa supernatants (Penner et al., 2016). This Pf4 phage-mediated inhibition was found to be dose dependent and could be alleviated by phage denaturation. This inhibition of Pf4 was more significant in preformed A. fumigatus biofilms than during biofilm formation. In contrast, Pf4 had no effect on planktonic conidia (Penner et al., 2016). These findings suggest that the site of phage action is specific to the extracellular matrix or hyphae (Reichhardt et al., 2015). Another two phages, Pf1 and fd, showed no inhibitory action against A. fumigatus. Pf4 attaches to A. fumigatus hyphae, and fungal inhibition may occur at the biofilm surface. The shorter phage Pf1 did not bind as extensively to A. fumigatus biofilms as did Pf4 and exhibited less efficient inhibition (Penner et al., 2016).
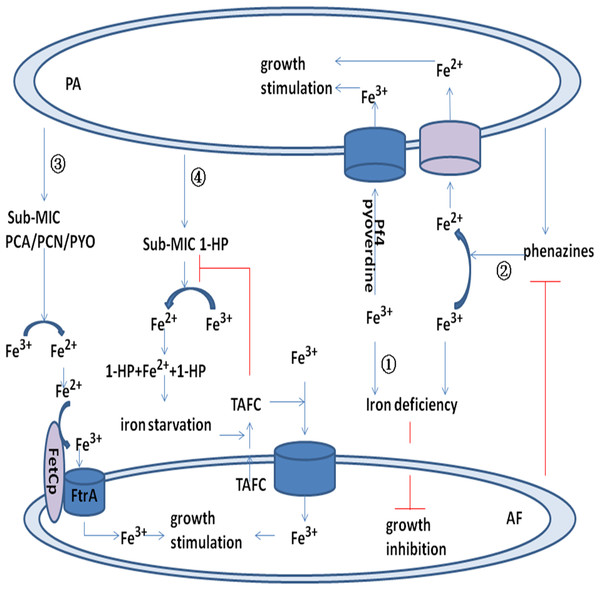
Figure 2: Model for the interaction between P. aeruginosa and A. fumigatus by iron uptake and competition.
Pyoverdine and Pf4 phage bind to Fe3+ and promote uptake by P. aeruginosa. Pyoverdine and Pf4 phage deprive A. fumigatus of Fe3+ and inhibit its growth (pathway ①). Phenazine reduces Fe3+ to Fe2+ and promotes P. aeruginosa uptake of Fe2+. Phenazine is converted by A. fumigatus into metabolic products with potentially modified redox potentials. These products may inhibit the reduction of Fe3+ in P. aeruginosa (pathway ②). Sub-MIC PYO, PCN, and PCA reduce Fe3+ to Fe2+ and promote the FetCp/FtrA complex of A. fumigatus to take up Fe2+ (pathway ③). Sub-MIC 1-HP reduces Fe3+ to Fe2+, and two 1-HP molecules bind the newly formed Fe2+. This chelating activity induces iron starvation and activates triacetylfusarinine C (TAFC). TAFC promotes A. fumigatus uptake of Fe3+ and stimulates its growth (pathway ④).Acute infection of P. aeruginosa by Pf bacteriophage can decrease the production of pyoverdine and the inhibitory capacity toward A. fumigatus biofilms. Thus, the reduced production of anti-microbials by P. aeruginosa infected by Pf bacteriophage may promote co-infection with A. fumigatus in CF airways (Secor et al., 2017).
SCVs in A. fumigatus intermicrobial competition
In a study of SCVs in intermicrobial competition with A. fumigatus (Anand et al., 2017b), the SCVs isolated from P. aeruginosa was shown to inhibit A. fumigatus biofilm formation, and this inhibitory capacity toward A. fumigatus biofilm was found to be related to pyoverdine (pathway ④ in Fig. 1) (Anand et al., 2017b). Indeed, isolated SCVs with high pyoverdine production had the highest inhibitory capacity in every co-culture method evaluated. Correspondingly, the two SCV isolates with the lowest inhibitory activities did not produce pyoverdine, suggesting that pyoverdine is the key P. aeruginosa inhibitor of A. fumigatus (Anand et al., 2017b).
Pseudomonas aeruginosa SCVs exhibit heterogeneity in inhibiting A. fumigatus biofilms. For instance, the inhibitory abilities of clinical SCVs isolates and reference CF non-mucoid isolates of P. aeruginosa or filtrates from P. aeruginosa planktonic or biofilm cultures were compared by coincubation with A. fumigatus during biofilm formation or in preformed biofilm. The metabolic activities of A. fumigatus biofilms were measured by different assays, with pyoverdine in filtrates being measured by spectrophotometry. The results showed that SCVs inhibited A. fumigatus biofilm formation, although the inhibitory effects of different SCVs were quite different. By adjusting planktonic culture filtrates, differences in SCV inhibition were related to SCV growth or deficient inhibitor production. Overall, the ability of SCVs to inhibit A. fumigatus biofilm was related to pyoverdine (Anand et al., 2017b). Thus, SCVs isolated from P. aeruginosa may be important in CF because they are capable of inhibiting A. fumigatus biofilm.
Inhibition of A. fumigatus by P. aeruginosa under conditions of hypoxia
Most studies examining the inhibition of A. fumigatus by P. aeruginosa have been performed under normoxic conditions. However, patients with acute exacerbation or progression of CF may exhibit hypoxia in focal lung sites (Cowley et al., 2015; Lambiase, Catania & Rossano, 2010; Worlitzsch et al., 2002). A. fumigatus inhibition by P. aeruginosa was recently evaluated under hypoxic conditions (Anand, Clemons & Stevens, 2017a), and the results showed that although the inhibitory activities of P. aeruginosa were effective under aerobic, hypoxic, or anaerobic conditions, P. aeruginosa growth was slow under hypoxic or anaerobic conditions, thus decreasing the ability of P. aeruginosa filtrates to inhibit A. fumigatus growth and biofilm formation. Regardless of the planktonic or biofilm state, the extracellular molecules produced by P. aeruginosa under anaerobic conditions were less inhibitory toward A. fumigatus growth and biofilm formation than were those under aerobic conditions. Therefore, the inhibitory power of P. aeruginosa against both A. fumigatus preformed biofilm and biofilm formation was decreased under hypoxic conditions (Anand, Clemons & Stevens, 2017a).
During the course of CF progression, P. aeruginosa mutants with a low level of pyoverdine production often appear, and the ratio of Fe3+ to Fe2+ decreases under hypoxic conditions. Intracellular iron acquisition by P. aeruginosa occurs mainly through the ingestion of low-activity Fe2+ via the production of phenazines and membrane permease (Cartron et al., 2006; Cornelis & Dingemans, 2013; Nguyen & Oglesby-Sherrouse, 2015). P. aeruginosa in the airway of CF patients typically shows low pyoverdine expression, and as mentioned above, pyoverdine can inhibit A. fumigatus growth by depriving A. fumigatus of iron. Under hypoxic conditions, the inhibitory effect of P. aeruginosa on A. fumigatus declines; thus, the growth of A. fumigatus is promoted (Sass et al., 2018). These findings explain why A. fumigatus is able to colonize CF airways following P. aeruginosa colonization and why A. fumigatus may persist during CF disease progression or chronic lung infection (Amin et al., 2010; Baxter et al., 2013a; Fillaux et al., 2012; Forsyth et al., 1988; Mirković et al., 2016; Nicolai et al., 1990; Speirs, Van Der Ent & Beekman, 2012).
After a long period of hypoxia, the interaction between P. aeruginosa and A. fumigatus appears to be similar to that under normoxic conditions (Anand, Clemons & Stevens, 2017a). Other factors may also affect P. aeruginosa and A. fumigatus interactions, such as prolonged use of antibiotics or inhaled corticosteroids (Noni et al., 2014).
The different inhibitory capacities of P. aeruginosa on A. fumigatus between planktonic and biofilm states
Pseudomonas aeruginosa and A. fumigatus are commonly found in the airways of patients with CF in the form of biofilms, and their pathogenicity and resistance in the biofilm state differ from those in the planktonic state. The inhibitory capacity of P. aeruginosa toward A. fumigatus in the biofilm state is also different from that in the planktonic state (Mowat et al., 2010). As an example, the supernatant extracted from P. aeruginosa biofilm was more effective than that extracted from planktonic cells (Ferreira et al., 2015). Pyoverdine plays an important role in P. aeruginosa biofilm formation, and its production is higher in biofilm than in planktonic cells (Visaggio et al., 2015). Thus, the P. aeruginosa biofilm-mediated suppression of A. fumigatus growth and biofilm formation is greater than planktonic P. aeruginosa-mediated suppression. It is also possible that other inhibitors may play an important role in this process (Anand et al., 2017b).
As mentioned above, the inhibitory effects of P. aeruginosa on A. fumigatus biofilm formation and preformed biofilms are different; it has been reported that P. aeruginosa can inhibit A. fumigatus biofilm formation but has almost no effect on preformed biofilms. The mature filamentous biofilms of A. fumigatus clearly restricted the inhibitory capacity of P. aeruginosa (Mowat et al., 2010), and another study showed that preformed A. fumigatus biofilm was more resistant to P. aeruginosa (Ferreira et al., 2015). According to a recent study, preformed A. fumigatus biofilms are inhibited by biofilm filtrates of P. aeruginosa strains isolated from CF patients via apoptosis, an effect that is related to mitochondrial membrane damage caused by metacaspase activation (Shirazi et al., 2016). In contrast, the inhibitory capacity of P. aeruginosa Pf4 phage toward A. fumigatus preformed biofilm is higher than that during biofilm formation. The P. aeruginosa phage Pf4 had little effect on planktonic conidial growth (Penner et al., 2016), suggesting that hyphae or the extracellular matrix is the specific site of phage action (Reichhardt et al., 2015).
The different inhibitory abilities of P. aeruginosa on A. fumigatus conidia and hyphae
After A. fumigatus conidia colonization in the CF patient airway, A. fumigatus gradually forms a biofilm that is rich in hyphae, and P. aeruginosa exhibits different inhibitory capacities toward A. fumigatus conidia and hyphae. In simultaneous static co-cultures, P. aeruginosa cells can effectively kill A. fumigatus conidia cells, but P. aeruginosa cells show only a minor inhibitory effect on sporelings grown for 12 h or longer as well as hyphae (Manavathu, Vager & Vazquez, 2014). Indeed, during co-cultivation with P. aeruginosa, A. fumigatus sporelings grown for 12 h or longer and young hyphae were stronger than ungerminated conidia with respect to the formation of P. aeruginosa–A. fumigatus biofilm.
Aspergillus fumigatus hyphae can withstand the fungicidal effect of P. aeruginosa and can produce the cytotoxic compound gliotoxin, which has anti-bacterial activity. Production of mycotoxin increases during mycelial growth and biofilm formation in A. fumigatus (Manavathu, Vager & Vazquez, 2014), and P. aeruginosa growth and its ability to kill A. fumigatus are suppressed with increasing levels of gliotoxin. In addition, virulence factor production and the inhibitory action of P. aeruginosa are strengthened by this increased metabolic activity in cells. Overall, the metabolic activity of germinating conidia and young sporelings is strong, whereas that of mature hyphae is limited in the apical regions of filaments. The apex, which has high metabolic activity, is the site at which P. aeruginosa binds to A. fumigatus hyphae and acquires nutrients (Toljander et al., 2007); hyphae, which has low metabolic activity, are not sensitive to the toxic molecules of P. aeruginosa. In fact, the cell walls of mature hyphae are poorly permeable to the toxic molecules of P. aeruginosa. Hence, mature hyphae are not easily killed by P. aeruginosa (Manavathu, Vager & Vazquez, 2014).
Comparison of the inhibitory effect between different P. aeruginosa strains from CF patients and non-CF patients
In addition to CF patients, P. aeruginosa and A. fumigatus also co-exist in the airways of patients with conditions such as chronic obstructive pulmonary disease, bronchiectasis, and hospital-acquired pneumonia. P. aeruginosa in CF patient airways can be divided into two types: mucoid and non-mucoid. A recent survey showed that both non-CF and CF A. fumigatus strains are inhibited by P. aeruginosa metabolic products (Nazik et al., 2017), and another study reported that both P. aeruginosa cells and filtrates isolated from CF patients had greater inhibitory effects on A. fumigatus growth and biofilm formation in vitro than did materials isolated from non-CF patients (Ferreira et al., 2015). Furthermore, non-mucoid P. aeruginosa exerted greater inhibitory effects on A. fumigatus than did mucoid P. aeruginosa in CF patients (Shirazi et al., 2016).
Pseudomonas aeruginosa isolated from the airway of CF patients has a greater chance of contact and longer duration of co-existence with A. fumigatus than isolates from non-CF patients. In addition, P. aeruginosa isolated from CF patients produces more toxic products and inhibitors than that from non-CF patients. Thus, CF patient-derived P. aeruginosa has a greater inhibitory effect against A. fumigatus growth and biofilm formation than strains isolated from non-CF patients. Mucoid P. aeruginosa usually exists in the deep and hypoxic zone of the lung (Gaspar et al., 2013; Pressler et al., 2006; Tramper-Stranders et al., 2012), and the synthesis of toxic products and inhibitors of P. aeruginosa is reduced under hypoxic conditions. Overall, the inhibitory capacity of mucoid CF P. aeruginosa filtrates is less than that of non-mucoid CF filtrates.
Promotion of A. fumigatus growth by P. aeruginosa
Pseudomonas aeruginosa induces A. fumigatus growth through the action of sub-bacteriostatic concentrations of phenazines and volatile organic compounds (VOCs). Pathogen reproduction requires iron ions, and phenazines promote Fe3+ reduction in CF patients infected with P. aeruginosa (Hunter et al., 2013). In the early stages of infection, host immune cell molecules, such as lactoferrin or transferrin, actively chelate Fe3+ ions and inhibit the growth of P. aeruginosa and A. fumigatus. However, phenazines can promote P. aeruginosa and A. fumigatus growth by iron acquisition, and P. aeruginosa phenazines can reduce Fe3+ to Fe2+ and liberate Fe3+ from host immune cells (Banin, Vasil & Greenberg, 2005; Hernandez, Kappler & Newman, 2004; Wang et al., 2011). P. aeruginosa produces VOCs during the course of infection and reproduction, and these molecules can be detected in sputum samples of CF patients infected with P. aeruginosa (Goeminne et al., 2012).
Promotion effect of P. aeruginosa on A. fumigatus colonization and growth
Under certain conditions, P. aeruginosa promotes the growth of A. fumigatus. Therefore, many CF patients are susceptible to infection with A. fumigatus after infection with P. aeruginosa (Paugam et al., 2010). One study hypothesized that P. aeruginosa infection promotes the evolution of A. fumigatus sensitization (Kraemer et al., 2006).
Another study reported reduced colonization of A. fumigatus after anti-infective treatment of P. aeruginosa in patients with acute exacerbation of CF, potentially because the bacterium protects A. fumigatus via immune factors and growth conditions (Baxter et al., 2013b). During acute exacerbation in CF patients, the colonization and growth of A. fumigatus may be related to the negative effects of P. aeruginosa on host lung function and immune defense. Resistance of P. aeruginosa biofilms to host immune responses contributes to the growth and multiplication of A. fumigatus (pathway ⑤ in Fig. 1) (Baxter et al., 2013a). By analyzing pre- and post-antibiotic sputum samples from adult CF patients, a study showed that intravenous antibiotics targeting P. aeruginosa during CF pulmonary exacerbations had a negative impact on the colonization and growth of A. fumigatus (Baxter et al., 2013b). Because P. aeruginosa contributes to the colonization and growth of A. fumigatus in CF patients, it is also possible that both microbes work together to combat host immune factors, resulting in increased infection and decreased pulmonary function. Nonetheless, their relationship may become competitive with growth, and P. aeruginosa inhibits A. fumigatus in various ways, as described above. Further research is required to verify the interdependence between these two organisms for survival within the airways of CF patients.
Sub-bacteriostatic concentrations of phenazines induce A. fumigatus growth
Regarding P. aeruginosa and A. fumigatus competitive growth, the former produces phenazines to inhibit the growth of the latter (Zheng, Keller & Wang, 2015a), yet one study demonstrated that sub-bacteriostatic concentrations of phenazines can induce A. fumigatus growth in specific situations. For instance, it was reported that sub-bacteriostatic concentrations of PYO, PCA, and PCN can induce A. fumigatus growth by promoting iron uptake. The redox function of 1-HP, which is capable of chelating iron ions and inducing iron starvation in A. fumigatus, can also promote A. fumigatus growth (pathway ① in Fig. 1) (Briard et al., 2015).
Aspergillus fumigatus can obtain iron resources through low-affinity ferrous iron uptake, high-affinity reductive iron uptake and siderophore-mediated iron uptake (Schrettl et al., 2004). Three sub-bacteriostatic phenazines, PYO, PCA, and PCN, can induce the growth of an A. fumigatus mutant lacking SidAp and siderophore biosynthesis under iron starvation conditions, and it was suggested that phenazines reduce Fe3+ to Fe2+ and promote the ferroxidase FetCp/permease FtrAp complex of the A. fumigatus mutant to take up Fe2+ (pathway ③ in Fig. 2) (Briard et al., 2015). Fusarinine C (FsC) and triacetylfusarinine C (TAFC) are two extracellular siderophores of A. fumigatus, while ferricrocin and hydroxyferricrocin are two intracellular siderophores of A. fumigatus (Haas, 2012). Sub-bacteriostatic 1-HP produced by P. aeruginosa promotes A. fumigatus growth by iron chelation and stimulation of TAFC secretion (pathway ④ in Fig. 2) (Briard et al., 2015).
VOCs promote A. fumigatus growth
A recent study showed that VOCs released by P. aeruginosa can promote A. fumigatus growth (pathway ⑥ in Fig. 1) (Briard, Heddergott & Latgé, 2016), and dimethyl sulfide was found to be a VOC with an enhancement effect on A. fumigatus, which is mediated by the gas phase. During sulfur starvation, A. fumigatus utilizes exogenous VOCs to promote growth, and in patients with CF, it is possible that P. aeruginosa promotes the colonization and growth of A. fumigatus by releasing VOCs, causing a rapid decline in lung function (Briard, Heddergott & Latgé, 2016). This result is consistent with the results of past clinical studies showing that P. aeruginosa and A. fumigatus co-infection resulted in decreased lung function (Amin et al., 2010; Baxter et al., 2013b).
Without direct contact, P. aeruginosa and A. fumigatus interact through signaling molecules such as VOCs. With direct contact, P. aeruginosa can release the corresponding signaling molecules to promote A. fumigatus colonization and growth. However, when these pathogenic microorganisms grow and contact one another, they exert a mutual inhibitory effect with regard to nutrient competition (Briard, Heddergott & Latgé, 2016).
The effect of A. fumigatus on P. aeruginosa
Pseudomonas aeruginosa and A. fumigatus interact with each other under co-growth conditions in patients with CF and other chronic pulmonary infection diseases. A. fumigatus can also resist inhibition by P. aeruginosa and, to a certain extent, affect its growth and metabolism.
Metabolic transformation of phenazines produced by P. aeruginosa
Phenazines inhibit A. fumigatus by inducing the production of ROS and RNS. Sod2p of A. fumigatus can resist the injury caused by ROS and RNS and antagonize inhibition by P. aeruginosa (Briard et al., 2015). Another study demonstrated that the phenazines produced by P. aeruginosa can be metabolically converted by A. fumigatus to reduce their inhibitory effect. As an example, PCA can be converted to 1-HP,1-methoxyphenazine and phenazine-1-sulfate. Although 1-HP has an inhibitory effect on A. fumigatus, 1-HP was also able to induce the production of the A. fumigatus siderophores TAFC and FsC (Moree et al., 2012). Regardless, previous experiments have shown that 1-HP inhibits bacterial siderophore biosynthesis (Dietrich et al., 2008). PCA induces the reduction of Fe3+ to Fe2+ for P. aeruginosa biofilm formation (Wang et al., 2011), and conversion of PCA by A. fumigatus may also decrease P. aeruginosa iron acquisition for metabolism and biofilm formation. Phenazine is converted by A. fumigatus into metabolic products with potentially altered redox potentials, and these products may inhibit Fe3+ reduction in P. aeruginosa (pathway ② Fig. 2). The phenazines PYO and PCA produced by P. aeruginosa can be converted to phenazine dimers by A. fumigatus, which have a decreased inhibitory effect on A. fumigatus. The QS signaling molecule PYO of P. aeruginosa affects transcriptional regulation and induces biofilm formation. Metabolic conversion of PYO by A. fumigatus might have an effect on QS system regulation. Thus, A. fumigatus can transform P. aeruginosa metabolites and radically alter the effects on their interaction, including the degree of inhibition (pathway ② in Fig. 1) (Moree et al., 2012).
Gliotoxin produced by A. fumigatus interferes with the metabolic growth of P. aeruginosa
During co-culture, A. fumigatus can also invoke its own metabolism and signaling molecules to disrupt P. aeruginosa growth. Gliotoxin produced by A. fumigatus is also a major immunoevasive toxin that is important in mediating A. fumigatus-associated colonization within the context of CF (Chotirmall et al., 2014). Overall, the gliotoxin secreted by A. fumigatus suppresses the inhibitory ability and growth of P. aeruginosa (pathway ⑦ in Fig. 1) (Manavathu, Vager & Vazquez, 2014).
Reducing the sensitivity of P. aeruginosa to antibiotics and promoting chronic infection
Under specific conditions, A. fumigatus can maintain the growth of P. aeruginosa in a co-existence scenario. One case-control study showed that CF patients infected by A. fumigatus could easily develop chronic P. aeruginosa infections despite receiving anti-microbial therapy (pathway ⑧ in Fig. 1) (Pressler et al., 2006).
The sensitivity of P. aeruginosa to antibiotics also changes when it co-exists with A. fumigatus. One in vitro study showed that P. aeruginosa in a polymicrobial biofilm with A. fumigatus was less susceptible to cefepime than P. aeruginosa in a monomicrobial biofilm state (pathway ⑧ in Fig. 1) (Manavathu, Vager & Vazquez, 2014). The extracellular matrix of polymicrobial biofilms differs for P. aeruginosa monomicrobial biofilms, and the sensitivity of P. aeruginosa in polymicrobial biofilm to some antibiotics decreases as a result of the change in the biofilm extracellular matrix. Indeed, some antibiotics cannot kill P. aeruginosa in biofilm due to altered permeability of the polymicrobial biofilm extracellular matrix. In contrast, the change in extracellular matrix between polymicrobial biofilms and A. fumigatus monomicrobial biofilms is not obvious and may not be sufficient to cause changes in the sensitivity of A. fumigatus to anti-fungal agents. For example, the sensitivity of A. fumigatus to anti-fungal drugs, such as voriconazole and posaconazole, did not change in polymicrobial or monomicrobial biofilms (Manavathu, Vager & Vazquez, 2014).
At present, only a few studies have examined the effect of A. fumigatus on P. aeruginosa, and the underlying mechanism should be the focus of further research.
Conclusion
In the co-infection state, P. aeruginosa interacts with A. fumigatus in a number of ways. In the early stage of CF, P. aeruginosa first colonizes and then grows, providing favorable nutritional and immunological conditions for infection and colonization by A. fumigatus. P. aeruginosa and A. fumigatus in the CF patient airway then promote each other’s growth and grow together. As the disease progresses and resources become less abundant in the co-existence environment, the interaction between P. aeruginosa and A. fumigatus shifts to mutual inhibition. It is suggested that antibiotic treatment against P. aeruginosa can inhibit the progression of A. fumigatus infection early during co-infection. When the infection is exacerbated and the condition of the CF patients deteriorates, anti-infective treatment against one pathogen may lead to the growth and reproduction of the other pathogen. At present, combined anti-infective therapy against both pathogens should be used, as the above possibilities need to be further investigated by a large number of clinical studies. The interaction between the two pathogens is quite complicated in the process of CF disease development, and many mechanisms of action remain unclear. Further studies examining the influence of A. fumigatus on P. aeruginosa and the immunomodulatory mechanism between these pathogens and the human body must be carried out to facilitate the treatment of CF patients with polymicrobial infections.