An online survey of personal mosquito-repellent strategies
- Published
- Accepted
- Received
- Academic Editor
- Giovanni Benelli
- Subject Areas
- Animal Behavior, Entomology, Public Health
- Keywords
- Survey, Mosquito, Repellents
- Copyright
- © 2018 Moore et al.
- Licence
- This is an open access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, reproduction and adaptation in any medium and for any purpose provided that it is properly attributed. For attribution, the original author(s), title, publication source (PeerJ) and either DOI or URL of the article must be cited.
- Cite this article
- 2018. An online survey of personal mosquito-repellent strategies. PeerJ 6:e5151 https://doi.org/10.7717/peerj.5151
Abstract
Background
Mosquito repellents can be an effective method for personal protection against mosquito bites that are a nuisance and carry the risk of transmission of mosquito-borne pathogens like plasmodia, dengue virus, chikungunya virus, and Zika virus. A multitude of commercially available products are currently on the market, some of them highly effective while others have low or no efficacy. Many home remedies of unknown efficacy are also widely used.
Methods
We conducted a survey study to determine what kind of mosquito repellents and other mosquito control strategies people use. Our online survey was focused on unconventional methods and was answered by 5,209 participants.
Results
The majority of participants resided in the United States, were female (67%), had higher education (81% had a university degree), and were 18 to 37 years old (50%). The most commonly used repellent was DEET spray (48%), followed closely by citronella candles (43%) and ‘natural’ repellent sprays (36%). We collected a plethora of home remedies and other strategies people use that warrant further research into their effectiveness.
Discussion
Our study lays the foundation for future research in alternative, unconventional methods to repel mosquitoes that may be culturally acceptable and accessible for people.
Background
Mosquitoes are vectors for infectious diseases that cause widespread epidemics and human morbidity (Brower, 2001). The pathogens transmitted by mosquitoes are quite diverse and include protozoans, arboviruses, and filarial nematodes (Fredericks & Fernandez-Sesma, 2014; Marquardt, 2004). These pathogens are taken up from one host and transmitted to another during the process of blood feeding. Mosquito-transmitted diseases put hundreds of millions of people at risk and still kill more than half a million people every year despite immense international efforts to combat them (Newby et al., 2016). Developing countries in tropical and subtropical regions bear the greatest burden with the majority of fatalities being young children (Snow et al., 2005).
The host-seeking behavior of mosquitoes as well as many other hematophagous arthropods depends heavily on their sense of smell. A battery of specific odorant receptors expressed in odorant receptor neurons within the antennae of mosquitoes enables them to detect a variety of chemical clues that are emitted by the host (Bohbot et al., 2007; Carey et al., 2010). Many of these attractants, for example CO2, organic acids, and aldehydes, have been identified and linked to specific receptor proteins (Beever, 2006; Chen & Luetje, 2014; Dong et al., 2013; Gopal & Kannabiran, 2013; Jones et al., 2012; Kumar et al., 2013; Nichols, Chen & Luetje, 2011; Suh, Bohbot & Zwiebel, 2014; Taylor et al., 2012; Turner et al., 2014; Xu et al., 2014). Olfactory receptor agonists as well as antagonists can impede the mosquito’s sense of smell and interrupt host-seeking behavior. Chemicals that elicit such responses are termed mosquito repellents.
Reducing the number of host-vector interactions is an effective way to reduce the spread of vector-borne diseases. Currently, only a small number of active ingredients in a large number of different commercially available formulations are widely used to protect humans from mosquitoes and other blood-sucking arthropods. DEET (N,N-Diethyl-meta-toluamide) is commonly used as an active ingredient in insect repellents (Hansen et al., 2014; Xu et al., 2014). Unsubstantiated fears of possible side effects of DEET have created a large market for “natural” DEET-free repellents with a variety of active ingredients. Picaridin, IR 3535, and a large assortment of essential oils, such as eucalyptus and lemongrass, are sold as sprays, creams, and integrated in wearable devices for repelling mosquitos (Xue, 2015). For a review of the long history and present use of plant extracts as commercial insecticides please see a recent review by Pavela (2016). A current text search on http://www.amazon.com with the search string “insect repellent” resulted in 22,950 hits.
Access to effective repellent products is often limited in developing countries. People with a high risk of vector-borne disease infections often have no or only insufficient means to protect themselves (Benelli & Mehlhorn, 2016). Across cultures, people use a variety of home remedies and traditional practices to repel mosquitos (Scandurra et al., 2014). Some of these remedies are likely highly effective, while others (Acheson, Plowright & Kerr, 2015; Clark et al., 2016; Scandurra et al., 2014) like DEET are expensive and scarce. In addition, some known effective methods, such as the use of mosquito nets, are impractical in particular situations and are therefore not used regularly by community members (Acheson, Plowright & Kerr, 2015; Clark et al., 2016; Scandurra et al., 2014). Several ethnobotanical research projects have focused on the traditional use of native plants as insect repellents (Pavela & Benelli, 2016; Tisgratog et al., 2016).
In a recent study, we have determined the efficacy of several commercial products, two fragrances, and a vitamin B patch in repelling mosquitoes (Rodriguez et al., 2015a ). The products were tested using a Y-tube olfactometer setup with Aedes aegypti (Linnaeus) and Aedes albopictus (Skuse), both major human disease vectors. Repellents with DEET or p-menthane-3,8-diol (PMD) as active ingredients had a prominent repellency effect over longer times and on both species. Some of the DEET-free products containing citronella or geraniol did not have any significant repellency effect. Interestingly, the perfume we tested had a significant repellency effect early after application. These findings were widely reported in the media and we received information from the public about other personal hygiene products that are used as mosquito repellents (see below).
In a follow up study, we performed attraction-inhibition assays using a taxis cage in a wind tunnel setting (Rodriguez et al., 2017). One person was placed upwind of the taxis cage and the mosquito movement towards or away from the person was recorded. The person was treated with various spray-on repellents or equipped with different ‘mosquito repellent devices’. We found that the spray-on repellents containing 98% DEET or 30% PMD had the highest efficacy in repelling mosquitoes compared to repellents with other ingredients. From the five wearable devices that we tested, only the one that releases Metofluthrin significantly reduced the numbers of attracted mosquitoes. A citronella candle had no effect. We concluded that many of the products that we tested that were marketed as repellents do not reduce mosquito attraction to humans.
The current study used an online survey to find unconventional methods for mosquito control and repellent practices that will be tested in future experiments. Here we define an unconventional method as one involving a product that is not commercially available worldwide, or involving products that are not used for their original purpose (i.e., dryer sheets). We also considered behaviors that are not obviously connected to mosquito control (i.e., drinking alcohol or eating bananas) as ‘unusual’. Links to the survey were distributed globally in an effort to capture a broad diversity of practices; however, outreach proved to be very complicated. The supplementary material shows the demographics of our study were biased; however, we did attain very useful qualitative input from our respondents.
Methods
Online survey
We developed a nine question online survey (see Supplemental Information 1). Demographic variables included gender, age, education levels and residence. Survey questions assessed people’s awareness of mosquitoes, general knowledge about mosquito-borne diseases and mosquito repellents and what mosquito control strategies people most frequently use. Two survey questions were specifically designed to gather unconventional mosquito repellent methods and strategies. The survey was available in English, Spanish, and Portuguese and was distributed in countries in which residents were likely to speak one of the above three languages. Convenience and snowball sampling were used.
Data collection
The data was collected anonymously through the online portal Survey Monkey (Palo Alto, CA, USA). Participants were recruited through various electronic means including postings on Facebook or other social media, and email list serves. The survey was shared on Facebook groups most of which were academics-led with ‘entomology’ or ‘health’ in the title, with the assumption that these groups are interested in this topic resulting in more participation and shares. The survey was also provided to various universities through personal contacts of the authors. The authors also forwarded the survey to their personal email contact lists. Those who agreed to participate had the option to enter a drawing for one of 10 gift cards valued between $50 and $100.
Data analysis
The data was exported into excel from Survey Monkey and categorized by manual inspection. For questions for which answers were provided, the Excel sort function was used to group and count identical answers. Answers to the open questions were analyzed by the authors and placed into different categories that we defined. Geographic region was determined by the continental location of the respondents’ current residence. Open-ended responses to the survey question regarding unconventional practices were categorized by type of mosquito control or repellent method.
Ethics statement
The study was approved by the NMSU Institutional Review Board (IRB # 15378, approved July 10th 2017). Consent was obtained by participants’ selecting to continue on to the survey after reading a description of the study, including risks and benefits.
Results
Demographics
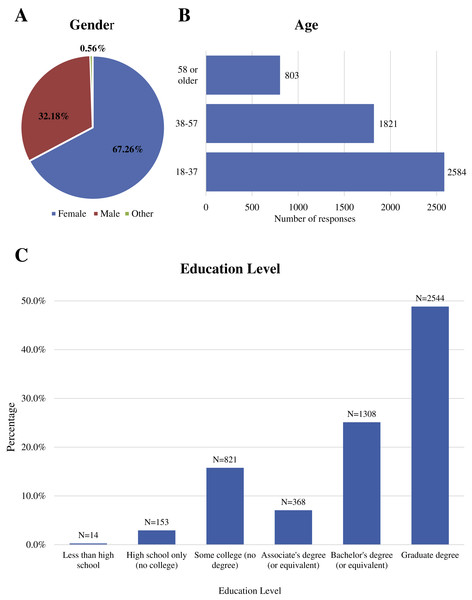
We received a total of 5,209 responses to our survey (for raw data, please see Supplemental Information 2). Figure 1 shows the demographics of the survey participants, where 67.71% of participants identified as female, 31.7% as male, and 0.59% as other; the majority of respondents were 18 to 37 years , followed by ages 38 to 57 years with ages over 58 years being the lowest (N = 789); and almost 50% held a graduate degree. Listed in Supplemental Information 3 are the regions and countries in which the respondents stated they were currently residing at the time of the survey. Over 76% were from North America, with 70% of the total participants listing the United States. Europe had the next highest number of responses, totaling to a little over 7% of total respondents followed by Australia (6.3%), Asia (4%), Africa (3.8%) and South America (3.3%).
Figure 1: Demographics of study participants.
(A) Gender of participants. (B) Age of participants in years. (C) Educational level of study participants.Mosquito repellent methods
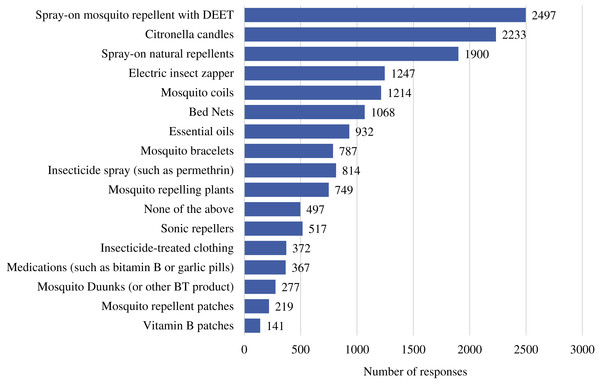
We chose 13 mosquito repellent/control methods and asked respondents to select all methods they had ever used. Answer choices can be seen in Fig. 2. Out of the 5,209 total respondents, 4,773 responded to the multiple choice question. The most common repellent method chosen by the respondents was spray-on mosquito repellent with DEET, closely followed by citronella candles and ‘natural’ spray-on repellents (Fig. 2). In the open ended question picaridin was mentioned by 59 participants and IR3535 only once.
Figure 2: Commonly used mosquito repellent methods.
The number of each response from one of the multiple choice questions.| Responses | Administration/preparation | Region | Ref. No. |
|---|---|---|---|
| Telephone app (sound) | Application | Sweden | |
| Cut down on sugar | Avoidance, Ingestion | USA, Denmark | |
| No meat consumption | Avoidance | UK | |
| Bananas | Avoidance, Ingestion, Topical, Unspecified | USA, Canada, New Zealand, Pakistan | Effiom, Avoaja & Ohaeri (2012) |
| Scented perfume/lotions/detergents | Avoidance, Topical | USA, Canada, Australia, Brazil, Switzerland, Ireland, China, Sri Lanka | Douglas (2008) |
| Animal dung (cow, elephant) | Burning | N. America, Australia, India | Lale & Kulkarni (2010) |
| Artemesia vulgaris (L.) (Asian mugwort) | Burning, Planting | USA | Liu et al. (2013), Tripathi et al. (2000) |
| Ayurvedic leaves | Burning | Sri Lanka | McPartland (1997) |
| Laurus nobilis (L.) (bay leaves) | Burning | USA | |
| Coconut husks | Burning | USA, Australia | Kulkarni (2017) |
| Coconut shell with dhuna | Burning | India | Kulkarni (2017) |
| Coffea Arabica (L.) (coffee) | Burning | Europe, Brazil, USA, Africa | Satho et al. (2015) |
| Larrea tridentate (de Candolle) (Creosote) | Burning | USA | Green, Beroza & Hall (1960) |
| Dhup-an | Burning | Bangladesh | Sharma, Chauhan & Lal (2005) |
| Dried Chrysanthemum spp. plants | Burning | Kenya | Isman (2006) |
| Dry Vitex negundo (L.) (Nochi) leaves | Burning | India | Arutselvi et al. (2012) |
| Dry powdered rhizomes of certain plants | Burning | India | Choochote et al. (2005) |
| Eucalyptus globulus (Labillardiere) (Tasmanian bluegum) leaves | Burning | Brazil, USA | Maia & Moore (2011) |
| Formes fomentarius (L.) (tinder fungus) | Burning | Sweden | |
| Poaceae (L.) (grass) | Burning | Canada | Yoon et al. (2015) |
| Lemon scented candles | Burning | Netherlands | Jaenson, Garboui & Pålsson (2006) |
| Prosopis juliflora (DC.) (mesquite) | Burning | USA | Tibbets & Faeth (1999) |
| Mixture of diesel fuel & malathion | Burning | USA | Abdel-Sattar et al. (2010) |
| Citrus sinensis (L.) (orange) peel or dried rind | Burning | Europe, Africa, USA, Canada Brazil | Amusan, Idowu & Arowolo (2005) |
| Citrus sinensis (L.) (orange) peel with Syzygium aromaticum (L.) (clove) | Burning | USA | Zhu et al. (2001) |
| Peat | Burning | Sweden | Kettridge et al. (2014) |
| Schinus terebinthifolius (Raddi) (pepper tree) | Burning | Namibia | Karunamoorthi, Ramanujam & Rathinasamy (2008) |
| Salvia officinalis (L.) (sage) | Burning | USA | Ikeura, Kobayashi & Hayata (2012) |
| Tamarix (L.) (salt cedar) | Burning | USA | Hagler & Buchmann (1993) |
| Santalum album (L.) (sandalwood) | Burning | Australia | Amer & Mehlhorn (2006) |
| Spices such as Cinnamomum (Schaffer) (cinnamon), Mentha (L.) (mint), Salvia officinalis (L.) (sage), Petroselinum crispum (Miller) (parsley) | Burning | USA | Ishii, Matsuzawa & Vairappan (2010) |
| Nicotiana tabacum (L.) (tobacco) or Cannabis sativa (L.) (marijuana) | Burning | USA | Gozan et al. (2014) |
| Vanilla candles | Burning | USA | |
| Cigar smoke | Burning, Topical | USA, Ireland, Hungary | Jufri, Irmayant & Gozan (2016) |
| Kerosene | Burning, Unspecified | USA, Bangladesh, New Zealand | Pates et al. (2002) |
| Brewer’s yeast | Ingestion | Canada, New Zealand, USA | Bisseleua, Gbewonyo & Obeng-Ofori (2008) |
| Capsaicin | Ingestion | USA | Rogers (1984) |
| Pelargonium citrosum (Citronella) tea | Ingestion | Brazil, Mexico (herbal tea) | Maia & Moore (2011) |
| Dill pickles and Allium sativum (L.) (garlic) | Ingestion | Unknown | |
| Gin and tonic | Ingestion | Chile, New Zealand, South Africa | |
| Zingiber officinale (Roscoe) (ginger) tea | Ingestion | USA | Zhang, McAuslane & Schuster (2004) |
| Citrus paradise (Macfad.) (grapefruit) juice | Ingestion | USA | Yoon et al. (2015) |
| Iron pills | Ingestion | USA | |
| Local honey | Ingestion | USA | |
| Marmite | Ingestion | New Zealand | Bisseleua, Gbewonyo & Obeng-Ofori (2008) |
| Citrus sinensis (L.) (oranges) | Ingestion | Canada, USA | Amusan, Idowu & Arowolo (2005) |
| Origanum vulgare (L.) (oregano) oil | Ingestion | Canada | Licciardello et al. (2013) |
| Selenium supplements | Ingestion | USA | Angradi & Tzilkowski (1987) |
| Tonic water with quinine | Ingestion | USA, South Africa, Malaysia, France, New Zealand, Australia | |
| Alcohol | Ingestion, Avoidance | USA, Canada, Denmark, Hungary, Ireland | Lefèvre et al. (2010) |
| Apple cider vinegar | Ingestion, Spray, Topical | USA, Europe | |
| Aloe vera | Ingestion, Topical | South Africa, Italy, Germany, Norway, Australia | Subramaniam et al. (2012) |
| Cinnamomum (Schaeff.) (cinnamon) | Ingestion, Topical, Unspecified | USA, South Korea, Canada | Amer & Mehlhorn (2006) |
| Car Mobil | Larvicide | India | |
| Charcoal tabs in standing water | Larvicide | USA | Belant et al. (1997) |
| Clorox | Larvicide | USA | Aubernon et al. (2015) |
| Coffee disposal granules | Larvicide, Spray | USA, Brazil | Satho et al. (2015) |
| Diesel | Larvicide, Spray | Egypt, USA | Leverkus et al. (2017) |
| Cut a Solanum lycopersicum (L.) (tomato) in half and leave it next to the bed | Other | South Africa | Bleeker et al. (2009) |
| Cut Syzygium aromaticum (L.) (lemon) | Other | USA, Malaysia | Fradin & Day (2002) |
| Hanging dried Eucalyptus globulus (Labill.) (eucalyptus) branches | Other | USA | Pavela & Benelli (2016) |
| Hanging Ziploc bag of water with penny or lavender inside | Other | USA | |
| Jar of sugar water away from people | Other | USA | |
| Keep cut Syzgium aromaticum (L.) (lemon) in all the rooms | Other | Malaysia | Fradin & Day (2002) |
| Tagetes minuta (L.) (Khakibos) under mattress. | Other | South Africa | Brown, Ainslie & Beinart (2013) |
| Moth balls | Other | USA | Harris, Palmer & George (1983) |
| Allium cepa (L.) (onion) by side of the bed | Other | USA | Ueno et al. (2003) |
| Pteridium aquilinum (L.) (Bracken fern) | Plant | Canada | Donnelly, Robertson & Robinson (2002) |
| Chrysathemum (L.) (chrysathemums) | Plant | USA | Kamaraj et al. (2011) |
| Eupatorium capillifolium (Lamarck) (dog fennel) | Plant | USA | Tabanca et al. (2010) |
| Equisetum (L.) (horse tail) | Plant | USA | Bunescu, Florian & BODIŞ (2012) |
| Monarda punctata (L.) (horsemint) | Plant | USA | Tabanca et al. (2013) |
| Lippia javanica (Musudzungwane) | Plant | South Africa | Kruger et al. (2015) |
| Achillea millefolium (L.) (yarrow) | Plant | Canada | Moore & Debboun (2007) |
| Tagetes (L.) (Marigolds) | Plant, Larvicide | USA, Canada | Pavela & Benelli (2016) |
| Solanum lycopersicum (L.) (tomato plant) | Plant, Other | Austria, Switzerland, South Africa | Bleeker et al. (2009) |
| Melissa officinalis (L.) (lemon balm) | Plant, Topical | USA | Amer & Mehlhorn (2006) |
| Rosmarinus officinalis (L.) (rosemary) | Plant, Topical, Burning | USA, Australia, Hungary, Canada | Isman (2006) |
| Nepeta cataria (L.) (catnip) | Plant, Topical, Other | USA | Alem & Douglas (2004) |
| Lavandula (L.) (lavender) | Plant, Topical, Spray, Unspecified | USA, Germany, Canada, Australia | Amer & Mehlhorn (2006) |
| Ocimum basilica (L.) (basil) | Plant, Topical, Unspecified | USA, Australia, Ireland | Del Fabbro & Nazzi (2008) |
| Pelargonium (L.) (geraniums) | Plants | USA, Switzerland, Canada, Eritrea | Tabanca et al. (2013) |
| Callicarpa americana (L.) (Beauty berry) | Plants, Topical, Unspecified | USA, UK | Cantrell et al. (2005) |
| Beer, mouthwash, Epsom salt solution | Spray | USA | Lefèvre et al. (2010) |
| Boric acid (1%) and 10% sucrose solution | Spray | USA | Gore & Schal (2004) |
| Coconut oil and Dettol disinfectant | Spray | New Zealand | Sritabutra & Soonwera (2013) |
| Pinol (Mexican version of pinsol) | Spray | USA | Aubernon et al. (2015) |
| Thymus vulgaris (L.) (thyme) leaf tea mixed with Pelargonium citrosum (L.) (citronella) oil | Spray | USA | Isman (2006) |
| Water soaked Fragaria ananassa (Duchene) (strawberry) | Spray | Brazil | Ceuppens et al. (2015) |
| Listerine/mouth wash | Spray, Topical, Unspecified | USA, South Africa, Canada | Alexander et al. (1962) |
| Avon Skin So Soft | Topical | USA, Europe, Australia, South Africa | Rodriguez et al. (2015a) |
| Baby oil | Topical | USA | Akbar et al. (2005) |
| Baby wipes | Topical | USA | Akbar et al. (2005) |
| Bounce dryer sheet | Topical | USA, Saudi Arabia | |
| Cigarette butts soaked in alcohol | Topical | USA | Mondal et al. (2015) |
| Syzygium aromaticum (L.) (clove) in alcohol solution | Topical | Brazil | Plarre et al. (1997) |
| Cocos nucifera (L.) (coconut) oil | Topical | USA, Australia, Denmark, UK | Das et al. (2003) |
| Cream with Melaleuca alternifolia (Maiden & Betche) (tea tree) oil, Eucalyptus globulus (Labill.) (eucalyptus) oil, and bees wax | Topical | USA | Yang & Ma (2005) |
| Crushed dried Carica papaya (L.) (papaya) leaves | Topical | USA | Rawani et al. (2012) |
| Crushed Hyptis suaveolens (L.) (pignut) | Topical | Australia | Yang & Ma (2005) |
| Crushed Mentha (L.) (mint) | Topical | USA, Australia | Karamaouna et al. (2013) |
| Crushed seeds of Lepidium sativum (L.) (chandrashura), Brassica nigra (L.) (black mustard), and Ricinus communis (L.) (castorbean) | Topical | USA | Barone & Frank (1999) |
| Deer tallow | Topical | Alaska | Fargione & Richmond (1991) |
| Deodorant | Topical | New Zealand, USA | Verhulst et al. (2016) |
| Diatomaceous earth | Topical | USA | Islam et al. (2010) |
| Diluted fabric softener | Topical | USA | |
| Fresh aromatic leaves of Myrtaceae (Juss.) plants | Topical | Australia | Yaghoobi-Ershadi et al. (2006) |
| Germix | Topical | USA | |
| Lemon/lime juice | Topical | USA, Pakistan, Denmark | Amer & Mehlhorn (2006) |
| Local bear bread (a tree fungus, pollypore) | Topical | Alaska | |
| Mixture of baby oil and Citronella oil | Topical | Australia | Maia & Moore (2011) |
| Mixture of fresh or dried Petroselinum crispum (Mill.) (parsley) and apple cider vinegar | Topical | Unknown | |
| Mixture of garlic juice and crushed peels of limes and lemons | Topical | USA | |
| Mixture of vodka and citronella, geranium, and other essential oils | Topical | USA | Sakulku et al. (2009) |
| Mud | Topical | USA, Canada, Australia | |
| Ananas comosus (L.)(pineapple plant) | Topical | Canada | |
| Shampoo (as repellent) | Topical | Mexico, Malaysia, Britain, USA | |
| South African camphor oil ointment, called Zambuk | Topical | South Africa | Schearer (1984) |
| SPF 30 sunscreen | Topical | USA | Schueller & Romanowski (2016) |
| Talcum powder | Topical | USA | Mehr, Rutledgei & Morales (1985) |
| Tanacetum vulgare (L.) (tansey) leaves | Topical | Canada | Schearer (1984) |
| Melaleuca alternifolia (Maiden & Betche) (tea tree) oil | Topical | Ireland, USA, Norway, France, Australia | Ahmad, Aslam & Mamat (2016) |
| Thiamine patch | Topical | USA | Dua, Pandey & Dash (2010) |
| Tiger balm | Topical | USA, Cambodia, Australia, Malaysia | Sarwar et al. (2017) |
| Vanilla extract | Topical | USA | |
| Vaseline | Topical | South Africa, USA | Lindqvist, Lindqvist & Tiilikkala (2008) |
| Azadirachta indica (Juss.) (neem) | Topical, Burning, Plant | India, USA, Bangladesh, Eritrea, Brazil, West Indies, Germany, Nigeria | Isman (2006) |
| Vicks Vaporub/menthol | Topical, Other | USA, Canada, Australia, South Africa, Namibia | Alankar (2009) |
| Mentha piperita (L.) (peppermint) lotion/oil | Topical, Other (camping tent) | USA, Unknown | Geetha & Roy (2014) |
| Mixture of baby oil, menthol, and Dettol | Topical, Spray, Unspecified | Australia, New Zealand | Bunker & Hirschfelder (1925) |
| Crushed Backhousia citriodora (F.Muell.) (lemon myrtle) or Eucalyptus globulus (Labill.) (eucalyptus) leaves | Topical, Spray, Unspecified | Australia | Maia & Moore (2011) |
| Vinegar/vinegar based solutions | Topical, Spray, Unspecified | USA | Rahmat et al. (2014) |
| Crushed up leaves of the Myrica cerifera (L.) (Wax Myrtle tree) | Topical, Unspecified | USA, Australia | Cilek, Hallmon & Johnson (2010) |
| Aleurites moluccanus (L.) (Kukui nut) oil | Topical, Unspecified | USA (Hawaii) | Nakayama & Osbrink (2010) |
| Rubbing alcohol | Topical, Unspecified | USA, South Korea | Govere et al. (2000) |
| Witch hazel | Topical, unspecified | USA, Canada | |
| Baking soda | Unspecified | USA | Beresford et al. (1996) |
| Citrus bergamia (Risso.) (bergamot) oil | Unspecified | USA | Peterson & Coats (2001) |
| Actaea racemose (L.) (black cohosh) | Unspecified | USA | |
| Plantago major (L.) (broadleaf plaintain) | Unspecified | Germany | |
| Broken liquid aspirin | Unspecified | USA | Alem & Douglas (2004) |
| Melaleuca cajuputi (Thomas Powell) (cajuput) oil | Unspecified | Indonesia | Amer & Mehlhorn (2006) |
| Calendula officinalis (L.) (English marigold) oil | Unspecified | Canada | Tavassoli et al. (2011) |
| Capsicum annuum (L.) (cayenne) | Unspecified | USA | Wimalaratne et al. (1996) |
| Chlorine | Unspecified | USA | Mathis & Quarterman (1953) |
| Copper pins | Unspecified | Germany | Becker, Oo & Schork (2015) |
| Diluted Raw Armor’s starch | Unspecified | USA | |
| Hairspray | Unspecified | USA | |
| Jasminum (L.) (jasmine) | Unspecified | Ireland, Spain | Amer & Mehlhorn (2006) |
| Key lime | Unspecified | USA | |
| Mixture of Cymbopogon (Kurt Sprengel) (lemongrass) oil, mouthwash, and Prunus dulcis (Mill.) (almond) oil | Unspecified | USA | Ansari et al. (2000) |
| Mixture of Listerine and witch hazel | Unspecified | USA | Ansari et al. (2000) |
| Mixture of Melaleuca alternifolia (Maiden & Betche) (tea tree) oil, apple cider vinegar, and water | Unspecified | USA | Di Campli et al. (2012) |
| Mixture of WD-40, camphor/phenol, and mineral oil | Unspecified | New Zealand, USA | |
| Nail polish | Unspecified | USA | Govere et al. (2000) |
| Oil paint | Unspecified | Pakistan | Kareru et al. (2010) |
| Pogostemon cablin (Blanco) (patchouli) oil | Unspecified | USA, Canada | Maia & Moore (2011) |
| Pinus sylvestris (L.) (pine) oil | Unspecified | USA | Maia & Moore (2011) |
| Pine tar | Unspecified | USA | Thorsell et al. (1998) |
| Sesamum indicum (L.) (sesame) oil | Unspecified | USA | Trongtokit et al. (2005) |
| Mentha spicata (L.) (spearmint) oil | Unspecified | USA | Ansari et al. (2000) |
| Sulphur | Unspecified | USA, Canada | Ferraro (1995) |
| Thieves oil | Unspecified | USA | Dua, Pandey & Dash (2010) |
| Toothpaste | Unspecified | USA | Geetha & Roy (2014) |
| Trader Joe’s Tea Tree Tingle | Unspecified | USA | Ahmad, Aslam & Mamat (2016) |
| Windex | Unspecified | USA | Miller (1983) |
Unconventional mosquito repellent methods
The major goal of this study was to collect information on unconventional mosquito repellent and control methods. In one of the last survey questions, participants were asked to list additional repellent methods that were not previously mentioned. Table 1 shows 167 of the unconventional methods that survey participants listed. The table is organized into the type of survey responses, followed by the application/preparation of the product mentioned, and the regions from where the participants that listed it reside at the time of the survey. The last column shows scientific references for this particular method. If the product mentioned was used in multiple ways, the most commonly listed application was listed first. In general, we documented the use of various plants, smokes, personal hygiene products, household chemicals, softeners, diets, supplements, and other behaviors. Within this collection of mosquito repellent strategies, the most commonly listed method was topical application of various substrates including personal hygiene products, plant rubs, and household chemicals. The second most listed unconventional strategy was the use of smoke produced by incineration of various materials encompassing specific local plants, tobacco products, and candles. Following the burning strategy was ingestion of various food items and supplements. Using live plants as a spatial repellent was the fourth most commonly listed method we documented as unconventional. Many of these plants are known as sources for essential oils.
For some items we documented several ways of use. For example, garlic was used either by ingestion, applying to the skin, or creating a spray that was used as an outdoor spatial repellent. In some cases we documented conflicting attitudes toward certain interventions; for example both the ingestion, as well as the avoidance of ingestion of bananas as well as alcohol were listed as mosquito-avoidance strategies.
One of the most reoccurring responses was the use of dryer sheets. Over 200 participants have used dryer sheets of some sort as a repellent device. Not listed as frequently as dryer sheets but having participants from a diversity of regions mention it, were products of the Neem tree (Azadirachta indica (Jussieu)) and coffee. Neem products were used by either burning or topically and their use was stated by people from Brazil, USA, India, Nigeria, Eritrea, and Bangladesh. Coffee was listed as either being burned or used grounded on lawns or as a larvicide. Coffee was listed by participants that resided in Greece, South Africa, Brazil, United Kingdom, Canada, or the United States. Burning sage and planting and/or topical application of catnip were listed only by respondents currently residing in the US.
Discussion
The data that we collected from this survey were acquired with the intent to find unconventional methods of mosquito control or repellency. We were able to identify 167 different methods.
Unconventional mosquito control methods
Some of the unconventional methods reported include Aloe vera (L.) and grapefruit, both of which have been previously studied. Aloe vera (L.) in combination with bio-control agent B. sphaericus has shown strong larvicidal effects against A. aegypti (L.) larvae (Subramaniam et al., 2012). Grapefruit oil was tested as a repellent against the adult rice weevil, Sitophilus oryzae (L.) ( Yoon et al., 2007). Other methods warrant efficacy tests such as key lime, sugar consumption, and iron pills. Many of these unconventional methods can be further analyzed in order to identify the chemical component(s) that make them a repellent such as analyzing the chemical ingredients in specific toothpastes or Windex for their repellency effects.
Many of our respondents used DEET and citronella candles which is interesting because DEET is a very effective mosquito repellent while citronella candles are not (Rodriguez et al., 2017; Rodriguez et al., 2015a). These popular methods could be used as controls in future studies. Picaridin and other commercial insect repellent products were mentioned but not as often as DEET and citronella.
The participants listed many different methods which were used at varying degrees in different regions. This may be due to the regional availability of certain mosquito repellent products or regional preferences for a certain method. It was unexpected to find a high number of participants from North America listing bed nets as a method that they’ve used. Interestingly, many of the North American bed nets users noted that they used them abroad.
Limitations of this study
Despite the fact that the survey was available in three different languages—English, Spanish, and Portuguese—the overwhelming numbers of answers were received from English-speaking survey respondents. A higher percentage—around 75%—of the survey respondents held some sort of academic degree (associates, bachelors, or graduate) (Fig. 1). Also, we received a total of 5,209 responses with 3,645 of those responses from people currently residing in the United States. Therefore, our respondent pool was strongly biased towards English-speaking academics residing in the US (see Supplemental Information 3). This suggests that we were unable to successfully plant “community-based seeds” both in the US and other countries in which the survey would be shared outside the academic community. The high number of female participants in this survey study could indicate that the topic of mosquito control appeals more to females than to males. An alternative explanation is that females are more likely to respond to online surveys in general which is supported by other studies like the one done by Sax, Gilmartin & Bryant (2003), where both paper and online surveys were more heavily answered by females than males.
Challenges and future directions
Survey research presents some challenges for fully understanding how alternative repellents are used ( Krosnick et al., 2015). Our survey responses tended to include ingredients rather than practices. Some responses may have left out specific actions or ingredients that respondents did not consider important. Future research may further explore these uses through collaboration with anthropologists and other field researchers who can observe and document actual practices as well as interview key informants with particular expertise in these alternative practices, including traditional healers and community elders. The intent of this study was to identify potential affordable, accessible, and culturally relevant mosquito control practices that can be made widely available to the public. However, one ethical issue that will need to be addressed in the future research is the protection of traditional knowledge and the potential for exploitation of that knowledge for commercial gain (Mugabe, 1999).
Availability, choice, use, and effectiveness of repellents and other mosquito avoidance strategies are also important variables for mathematical models predicting mosquito-borne disease dynamics. The concept of ‘One Heath’ integrates human, animal and environmental health. This multidisciplinary approach is relevant for veterinarians, ecologists, biologists, and others to understand and predict the spread of disease among people, animals, and within the environment (Destoumieux-Garzón et al., 2018). One Health approaches will benefit from solid knowledge on local repellent use, and its efficacy. Thus, knowing how different people in different regions defend themselves against mosquitos, and knowing the efficacy of these methods, can be combined with other data to contribute to disease outbreak predictions (Benelli & Duggan, 2018).
In conclusion, we identified several understudied mosquito repellent methods that may or may not be more effective and practical across diverse global settings. In many cases the efficacy of these methods has not been evaluated in scientific experiments. This study offers opportunities for further research into unconventional mosquito repellent methods to determine their effectiveness. Those that prove to be effective may be taken up more quickly by populations at risk for contracting mosquito-borne diseases because local populations may have better access to them and they may fit better into the context of people’s daily lives.
Supplemental Information
Raw data
This file contains the individual answers of the survey participants.
Geographic distribution of all survey responses
This file shows the number of responses from all the countries from which there were responses.