Antimicrobial and antibiofilm activity of specialized metabolites isolated from Centaurea hyalolepis
- Published
- Accepted
- Received
- Academic Editor
- Liang Wang
- Subject Areas
- Biochemistry, Microbiology, Plant Science
- Keywords
- Multidrug resistant infections, Plant metabolites, Bio-guided purification, Antimicrobial natural compounds, Sesquiterpene lactones, Cnicin, Antibiofilm activity
- Copyright
- © 2024 Ismail et al.
- Licence
- This is an open access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, reproduction and adaptation in any medium and for any purpose provided that it is properly attributed. For attribution, the original author(s), title, publication source (PeerJ) and either DOI or URL of the article must be cited.
- Cite this article
- 2024. Antimicrobial and antibiofilm activity of specialized metabolites isolated from Centaurea hyalolepis. PeerJ 12:e16973 https://doi.org/10.7717/peerj.16973
Abstract
The discovery of plant-derived compounds that are able to combat antibiotic-resistant pathogens is an urgent demand. Over years, Centaurea hyalolepis attracted considerable attention because of its beneficial medical properties. Phytochemical analyses revealed that Centaurea plant species contain several metabolites, such as sesquiterpene lactones (STLs), essential oils, flavonoids, alkaloids, and lignans.The organic extract of C. hyalolepis plant, collected in Palestine, showed significant antimicrobial properties towards a panel of Gram-negative and Gram-positive bacterial strains when the Minimal Inhibitory Concentration (MIC) values were evaluated by broth microdilution assays. A bio-guided fractionation of the active extract via multiple steps of column and thin layer chromatography allowed us to obtain three main compounds. The isolated metabolites were identified as the STLs cnicin, 11β,13-dihydrosalonitenolide and salonitenolide by spectroscopic and spectrometric analyses. Cnicin conferred the strongest antimicrobial activity among the identified compounds. Moreover, the evaluation of its antibiofilm activity by biomass assays through crystal violet staining revealed almost 30% inhibition of biofilm formation in the case of A. baumannii ATCC 17878 strain. Furthermore, the quantification of carbohydrates and proteins present in the extracellular polymeric substance (EPS) revealed the ability of cnicin to significantly perturb biofilm structure. Based on these promising results, further investigations might open interesting perspectives to its applicability in biomedical field to counteract multidrug resistant infections.
Introduction
Finding healing powers in plants is an ancient idea. Plants have always represented an important source of therapeutic compounds and have inspired the production of synthetic or biotechnological drugs, including antimicrobial compounds. Recently, several efforts have been also devoted to the discovery of plant-derived compounds, which are able to counteract the worrying phenomenon of the spread of antibiotic-resistance (Machado et al., 2023). Antimicrobial resistance (AMR) is the ability of microorganisms to adapt and thrive in the presence of compounds previously able to influence their survival (Bottery, Pitchford & Friman, 2021). AMR is a worrying phenomenon threatening human health that, if not resolved, might significantly reduce the effectiveness of conventional antibiotics against bacterial infections (Álvarez-Martínez, Barrajón-Catalán & Micol, 2020). Bacteria are able to adapt to the presence of antibiotics by mutations, horizontal gene transfer or modifications of gene expression. Once antibiotic resistance is acquired, it could be transmitted to future bacterial generations either by cell division or by horizontal gene transfer (Christaki, Marcou & Tofarides, 2020). Antibiotic abuse in health sector, agricultural practices, together with the high rate of mobility of people, animals, and goods around the world have greatly contributed to the spread of antimicrobial resistance in recent years (Bottery, Pitchford & Friman, 2021). Considering the dearth of discovery of new antimicrobial drugs, the phenomenon has emerged as a grave threat to current and future medical practice and socioeconomic activity, as recently highlighted by numerous organizations and governments (Sulis, Sayood & Gandra, 2022). In this scenario, the development of effective alternatives to conventional antibiotics has become urgent and medicinal plants might represent a potential source of novel drugs.
A microbial community of cells adhered to an abiotic or a biotic surface, which is made of complex extracellular polymeric substance (EPS) composed primarily of proteins, polysaccharides, and eDNA is known as biofilm (Gandhi et al., 2017). The formation of bacterial biofilms is particularly due to interactions of filamentous bacteria, microbial aggregates, inorganic and organic particles, which are held together by EPS (Palomares-Navarro et al., 2023). Furthermore, the biofilm serves as a barrier to invasions and offers resistance towards protozoans, grazers, and host immunological responses (Gandhi et al., 2017). The formation of bacterial biofilm is also responsible for the ineffectiveness of conventional antibiotics (Cangui-Panchi et al., 2023). Indeed, the three-dimensional network of the EPS matrix protects bacteria from the effects of disinfectants, thus making biofilms difficult to eradicate (Flemming et al., 2016). Therefore, it is necessary to investigate and to explore new anti-biofilm compounds. In the last years, there has also been a growing application of biochemical and molecular biology methods to the isolation and purification of compounds with specific and potentiated activities. This requires several steps, such as extraction, fractionation, purification, characterization, and evaluation of biological activity and of putative cytotoxic effects (Cook et al., 2023).
Centaurea hyalolepis is among the plants that attracted attention for their therapeutic properties. It belongs to the well-known Asteraceae, also named Compositae family (Danin, 2018), one of the biggest plant families comprising several genera and more than twenty-three thousand species of shrubs, herbs, and trees (Danin, 2018; Djeddi, Argyropoulou & Skaltsa, 2008). The species belonging to the Centaurea genus are predominately distributed in West Asia and Mediterranean area (Tiwana et al., 2021). Since the phytochemical constituents of plants represent a precious reservoir of novel, diverse and potent drugs (Chemsa et al., 2018), researchers devoted several efforts to the investigation of wild plants endowed with beneficial medical properties. Centaurea species have been widely used as a remedy for several health-related issues, such as diarrhea, urinary tract infections, diuretic problems, diabetes, inflammation, bacterial infections, dandruff, and others (Chemsa et al., 2018; Zengin et al., 2010; Csupor et al., 2010; Shoeb et al., 2005). Phytochemical analyses revealed that Centaurea plant species contain several metabolites, such as sesquiterpene lactones (STLs), essential oils, flavonoids, alkaloids, and lignans (Khammar & Djeddi, 2012; Ismail et al., 1986). It is worth noting that STLs and flavonoids are the predominant metabolites of Centaurea (Maos, Anastácio & Nunes, 2021), and they are probably responsible for most of the reported biological activities of Centaurea crude extracts (Khammar & Djeddi, 2012).
Recently, STLs were widely analyzed for their biological properties, and found to confer antibacterial, antifungal, anti-inflammatory, anti-cancer, and anti-oxidative properties (Laurella et al., 2022). This elicited a great interest that led to the purification and isolation of STLs from Asteraceae family members which are known to be very rich in STLs (Picman, 1986). It was demonstrated that the unique bioactivity of STLs is correlated to their molecular structure characterized by the presence of α, β-unsaturated carbonyl groups with α-methylene or γ-lactone rings aside by the basic 15-carbon backbone structure (Picman, 1986; Kupchan et al., 1970). The molecular structure of STLs might allow covalent interactions with sulfhydryl groups of enzymes or other nucleophilic biological molecules (Kupchan et al., 1970; Schmidt, 1999). In this study, we found that C. hyalolepis crude extracts showed significant antimicrobial properties towards Gram-negative and Gram-positive bacterial strains, such as Staphylococcus aureus ATCC 29213, methicillin-resistant Staphylococcus aureus MRSA WKZ-2, Enterococcus faecalis ATCC 29212, Escherichia coli ATCC 25922, Salmonella enterica subsp. enterica Serovar Typhimurium ATCC 14028, and Acinetobacter baumannii ATCC 17878. Since dichloromethane (CH2Cl2) organic extract exhibited the strongest antibacterial activity with respect to other extracts obtained in different solvents, it was used as starting point to perform a bio-guided purification of C. hyalolepis extract. At the end of each step of the purification process, the obtained fractions were tested for their antimicrobial properties to select those containing plant metabolites responsible for the observed antibacterial activity. This manuscript reports for the first time the antimicrobial activity of C. hyalolepis plant extract and the isolation of three main metabolites by bio-guided purification as well as a significant antibiofilm activity of cnicin towards the tested bacterial strains. Compounds were identified by spectroscopic and spectrometric analyses and their stereochemistry was confirmed by comparing the optical rotation values with literature data.
MATERIALS & METHODS
Materials
A Jasco P-1010 digital polarimeter (Tokyo, Japan) was used to measure optical rotations. 1H NMR spectra were recorded at 500 MHz in CDCl3 and CD3OD on a Bruker spectrometer and the same solvents were used as internal standards. Electrospray ionization mass spectra (ESIMS) were obtained as previously described (Ismail et al., 2023). Column chromatography (CC) was performed on silica gel (Kieselgel 60, 0.063–0.200 mm; Merck, St. Louis, MO, USA). Preparative and analytical thin-layer chromatography (TLC) procedures were performed as previously described (Zorrilla et al., 2023). CC silica gels were from Merck (St. Louis, MO, USA), Kieselgel 60, 0.063–0.200 mm. All the reagents and the solvents were purchased from Merck (St. Louis, MO, USA), unless specified otherwise.
Bacterial strains and growth conditions
Six bacterial strains were used to evaluate the antibacterial properties of the plant extracts, fractions, and pure compounds. Tested bacterial strains include Staphylococcus aureus ATCC 29213, methicillin-resistant Staphylococcus aureus MRSA WKZ-2, Enterococcus faecalis ATCC 29212, Escherichia coli ATCC 25922, Salmonella enterica subsp. enterica Serovar Typhimurium ATCC 14028, and Acinetobacter baumannii ATCC 17878. The same strains were used for the biofilm tests. Bacterial strains were grown in Muller Hinton Broth (MHB; Becton Dickinson Difco, Franklin Lakes, NJ, USA) and on Tryptic Soy Agar (TSA; Oxoid Ltd., Hampshire, UK). In all the experiments, bacteria were inoculated and grown overnight in MHB at 37 °C (Gaglione et al., 2019).
Plant collection and identification
C. hyalolepis leaves were collected in West-Bank, Palestine, during March 2021 and identified by Dr. Ghadeer Omar (Department of Biology and Biotechnology at An-Najah National University in Palestine) by using “Field guide to wild flowers of Jordan and neighboring countries Vegetation of Israel and Neighboring Countries” (Al-Eisawi, 1998; Danin, 2018). A representative plant specimen has been deposited at An-Najah National University herbarium (voucher number ANUH1625). The collected leaves of C. hyalolepis were then washed with water, dried, and powdered using a blender prior to the extraction process.
Plant extraction
To prepare plant extracts, leaves powder (728.0 g) was macerated (1 × 2.5 L) for 48 h in H2O/MeOH (1/1, v/v) under stirring at room temperature. After centrifugation at 7.000 rpm for 40 min, the supernatant was extracted in n-hexane (3 × 1.0 L) and successively in CH2Cl2 (3 × 1.0 L). Methanol was then removed under reduced pressure and the residual water phase extracted by using EtOAc (3 × 500 mL).
Bio-guided fractionation and identification of isolated compounds
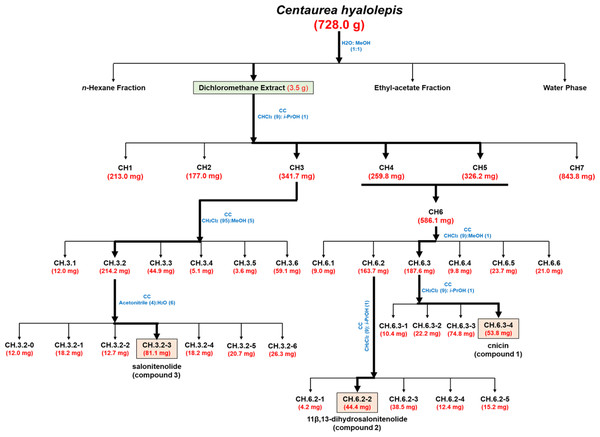
The organic extract obtained in CH2Cl2 (3.5 g), which showed antimicrobial activity against Gram-negative and Gram-positive bacterial strains (Table 1), was purified by CC performed using as eluent mixture a solution of CHCl3/i-PrOH (9/1, v/v) obtaining homogeneous fractions (Fig. 1). The preliminary antimicrobial activity was tested against two bacterial strains, i.e., the Gram-negative Salmonella enterica subsp. enterica Serovar Typhimurium ATCC 14028 and the Gram-positive E. faecalis ATCC 29212. In the case of antimicrobial activity, further analyses were performed against four additional bacterial species, such as S. aureus ATCC 29213, methicillin-resistant S. aureus MRSA WKZ-2, E.coli ATCC 25922, and A. baumannii ATCC 17878. Fractions CH.3-CH.5 were found to exert a significant antibacterial activity and were further purified as shown in Fig. 1. In particular, fractions CH.4 and CH.5 were combined into one fraction named CH.6 as they shared the same profile on TLC and showed similar antibacterial properties (Table 2). The remaining part of fraction CH.3 (341.7 mg) was further purified by CC and eluted in CH2Cl2/MeOH (95/5, v/v). Subsequent reverse-CC was eluted with acetonitrile/H2O (4/6, v/v) and allowed us to obtain a main metabolite (CH.3.2-3) identified as salonitenolide (compound 3, 81.1 mg). The remaining part of fraction CH.6 (568.1 mg) was firstly purified by CC and eluted with CHCl3/MeOH (9/1, v/v), thus allowing to obtain six homogeneous fractions (CH.6.1–CH.6.6). The remaining part of sub-fraction CH.6.3 (187.6 mg) was further purified by CC and eluted in CH2Cl2/i-PrOH (9/1, v/v), thus providing a main metabolite (CH.6.3-4) identified as cnicin (compound 1, 53.8 mg). The remaining part of the sub-fraction CH.6.2 (163.7 mg) was further purified by CC and eluted with CH2Cl2/i-PrOH (9/1, v/v) to obtain a pure sesquiterpene lactone identified as 11β,13-dihydrosalonitenolide (compound 2, 44.4 mg).
| MIC100(mg/mL) | ||||
|---|---|---|---|---|
| Bacterial strains | n-Hexane extract | CH2Cl2 extract | EtOAc extract | Residual water phase |
| S. aureus ATCC 29213 | 1 | 0.25 | 1 | >2 |
| S. aureus MRSA WKZ-2 | 1 | 0.5 | 1 | >2 |
| E. faecalis ATCC 29212 | 2 | 2 | 1 | >2 |
| E. coli ATCC 25922 | 2 | 1 | 1 | >2 |
| S. Typhimurium ATCC 14028 | 1 | 1 | 1 | >2 |
| A. baumannii ATCC 17878 | 2 | 0.25 | 0.5 | >2 |
Notes:
Figure 1: Bio-guided fractionation of the extracts.
Schematic overview of the successive bio-guided fractionation steps of the active extract obtained in CH2Cl2 solvent starting from C. hyalolepis leaves ending up with the pure isolated compounds.Antibacterial activity assays
Total extracts and compounds 1–3 were tested to evaluate their antimicrobial activity as previously described (Pizzo et al., 2018). Briefly, bacterial cells were diluted to 2 × 106 CFU/mL in Nutrient Broth (NB; Difco, Becton Dickinson, Franklin Lakes, NJ, USA) along with increasing amounts of each tested extract or compound. In each case, starting from a stock solution, two-fold serial dilutions were prepared according to broth microdilution method. Following drying, the isolated compounds and fractions were quantified by determining their dry weight by using a digital analytical balance. Stock solutions were prepared in DMSO and further diluted to achieve final DMSO concentration lower than 5%. After overnight incubation of bacterial cells with tested compounds, MIC100 values were determined as the lowest concentration responsible for no visible bacterial growth. At least three biological replicates were performed for each experiment.
Bactericidal activity assays
A colony-counting assay (Wiegand, Hilpert & Hancock, 2008) was used to determine the minimal bactericidal concentration (MBC) values of isolated compounds. To do this, bacterial aliquots were taken from the wells with no sign of bacterial growth. Samples were then serially diluted in 0.5X NB, plated on TSA (Tryptic Soy Agar) plates, and incubated for 24 h at 37 °C. Following incubation, bacterial colonies were counted, and the MBC values were determined as the lowest compound concentration that resulted in >99.9% cell death relative to the original bacterial inoculum (Dell’Olmo et al., 2021). The experiment was performed in triplicate.
| MIC100(mg/mL) | ||
|---|---|---|
| CH2Cl2Fraction | E. faecalisATCC 29212 | S. TyphimuriumATCC 14028 |
| CH.1 | 0.125 | 0.125 |
| CH.2 | 1 | 1 |
| CH.3 | 0.125 | 0.0312 |
| CH.4 | 0.125 | 0.0625 |
| CH.5 | 0.25 | 0.0312 |
| CH.7 | 1 | 1 |
Notes:
Antibiofilm activity assays
Biomass assays through crystal violet staining were used to evaluate the antibiofilm activity of cnicin (1). In particular, bacteria inocula were grown over-night at 37 °C, then diluted to 2 × 108 CFU/mL in 0.5X MHB containing increasing concentrations of cnicin (1) (0–1 mg/mL) and successively incubated at 37 °C for 24 h. Then the bacterial biofilm was washed three times with phosphate buffer (PBS 1X) and incubated for 20 min with 0.04% crystal violet at room temperature. Samples were washed with PBS and then the dye bound to the cells was dissolved in 33% acetic acid. A wavelength of 630 nm and a microtiter plate reader (FLUOstar Omega, BMG LABTECH, and Germany) were used for spectrophotometric measurements (Gaglione et al., 2017). The absorbance values at 630 nm were compared with those obtained for the untreated biofilm sample, thus obtaining the reported percentage of biofilm mass. Three biological replicates were performed for each experiment that was carried out with triplicate determinations.
Determination of total protein content in biofilm matrix
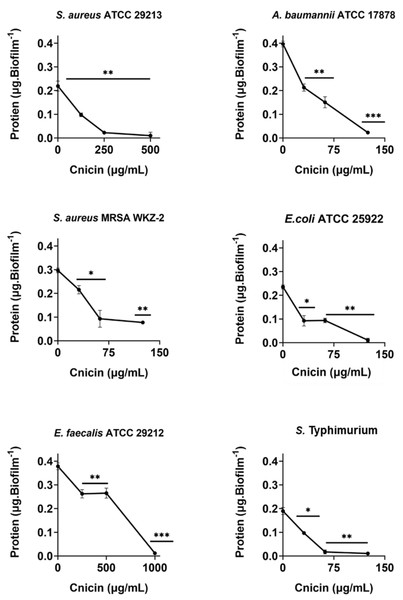
The effects of compound 1 on the total protein content of biofilm matrix were evaluated on the bacterial strains that were incubated with increasing concentrations of selected compounds (0–1 mg/mL, 1:1 v/v). Following incubation, protein content was evaluated by Bradford assay (Bradford, 1976). To do this, bacteria were grown overnight in MHB at 37 °C, and then adjusted to 4 × 108 CFU/mL in a final volume of 100 µL of 0.5X MHB (Difco, Becton Dickinson, Franklin Lakes, NJ) along with increasing concentrations of cnicin (compound 1) (1:1 v/v). The samples were then incubated at 37 °C for 24 h, in order to test compound 1 effects on protein content of formed biofilms. Biofilms were then harvested and suspended in 36 µL of Milli-Q water through ultrasonic oscillation. Afterwards, equal volumes of supernatant solution and Bradford Dye Reagent were mixed at room temperature for 5 min prior to measuring absorbance values at 595 nm (Guo et al., 2019). Protein amount was determined by using a standard bovine serum albumin (BSA, 0–2 mg/mL) calibration curve (Fig. S1). Three biological replicates were performed for each experiment.
Determination of total polysaccharides content in biofilm matrix
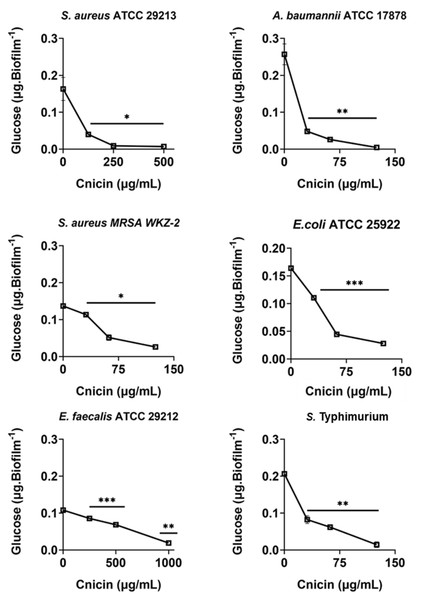
The antibiofilm effects of compound 1 were deepened by quantifying the polysaccharides content of biofilm matrix produced in the absence or in the presence of compound 1 by the six bacterial strains used in this study. To do this, phenol-sulphuric acid method was used (Nielsen, 2010). Bacteria were grown overnight in MHB at 37 °C, and then adjusted to 4 × 108 CFU/mL in a final volume of 100 µL of 0.5X MHB (Difco, Becton Dickinson, Franklin Lakes, NJ) along with increasing concentrations of cnicin (compound 1) (0–1 mg/mL, 1:1 v/v). The samples were then incubated for 24 h at 37 °C, to evaluate the effects of compound 1 on polysaccharides content of formed biofilms. Biofilms were then harvested, suspended in 36 µL of Milli-Q water and sonicated for 5min. Afterwards, equal volumes of 5% phenol and five volumes of concentrated H2SO4 were added to the samples that were incubated at room temperature for 30 min prior to measuring absorbance values at 482 nm (Guo et al., 2019). Polysaccharides amount was determined by using a standard glucose (0–5 mg/mL) calibration curve (Fig. S2). Three biological replicates were performed for each experiment.
Statistical analyses
Statistical analyses were performed by using a student’s t-test. Significant differences were indicated as *P < 0.05, **P < 0.01 or ***P < 0.001. Graphs were performed with the GraphPad Prism 8 software.
Results
C. hyalolepis leaves extraction, bio-guided fractionation of CH2Cl2 extract, and antimicrobial activity evaluation
C. hyalolepis leaves were extracted as described in Materials and Methods section. In particular, the hydro-alcoholic solution obtained by maceration has been extracted using solvents with increasing polarity, such as n-hexane, dichloromethane (CH2Cl2), and ethyl acetate (EtOAc). Gram-negative (E. coli ATCC 25922, A. baumannii ATCC 17878, and Salmonella enterica subsp. enterica Serovar Typhimurium ATCC 14028) and Gram-positive (S. aureus ATCC 29213, S. aureus MRSA WKZ-2, E. faecalis ATCC 29212) bacterial strains were employed to test the activity of the obtained organic extracts by using the broth microdilution assays (Wiegand, Hilpert & Hancock, 2008) to determine the minimal inhibitory concentration (MIC) values reported in Table 1.
The CH2Cl2 extract was found to exhibit the strongest antimicrobial activity against all the tested bacterial strains, with MIC100 values ranging from 0.25 to 2 mg/mL (Table 1). Thus, it was selected as a starting point to perform a bio-guided fractionation with the main aim to identify plant metabolites responsible for the observed antibacterial activity. To this purpose, CH2Cl2 extract was fractionated by CC as reported in Fig. 1 yielding six homogenous fraction groups, which were tested on two bacterial strains, i.e., Salmonella enterica subsp. enterica Serovar Typhimurium ATCC 14028 and E. faecalis ATCC 29212.
These two bacterial strains were selected since they were the most resistant to CH2Cl2 extract antibacterial activity (Table 1, MIC100 values ranging from 2 to 1 mg/mL), thus representing the prototype of a Gram-positive and a Gram-negative bacterial strain suitable to identify the active compounds present in this organic extract. As reported in Table 2, the fractions obtained upon purification by column chromatography were found to be more active than the total CH2Cl2 extract, being effective on both E. faecalis ATCC 29212 and Salmonella enterica subsp. enterica Serovar Typhimurium ATCC 14028 bacterial strains with MIC100 values comprised between 0.0312 and 1 mg/mL (Table 2). Since the two fractions CH.4 and CH.5 were found to share similar chromatographic profiles on TLC, they were combined to form fraction CH.6 which was further fractionated to obtain active metabolites.
Isolation and identification of pure metabolites from CH2Cl2 extract
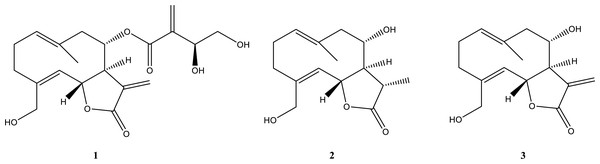
The most active fractions (CH.3-CH.5) were further purified by successive steps of CC (Fig. 1) obtaining three pure metabolites identified by spectroscopic and spectrometric methods (1H NMR and ESI MS) as cnicin, 11 β,13-dihydrosalonitenolide, and salonitenolide (compounds 1–3 in Fig. 2). The yields in percentage of compounds 1, 2, and 3 were found to be 1.5%, 1.2%, and 2.3%, respectively.
Figure 2: Isolated compounds.
Chemical structures of cnicin (compound 1), 11β,13-dihydrosalonitenolide (compound 2), and salonitenolide (compound 3).In particular, the 1H NMR and ESI MS data of compound 1 (Figs. S3 and S4) agreed with those reported in the literature for cnicin (Suchý et al., 1960). Its absolute configuration was confirmed comparing the specific optical rotation value [α]25D = +152.4 (c 0.5, MeOH) with that reported in the literature for this compound (Kurita et al., 2016). The structure and absolute stereochemistry of 11β,13-dihydrosalonitenolide (compound 2) was confirmed by comparing its 1H NMR and ESI MS data (Figs. S5 and S6) and the specific optical rotation value [α]25D = +81 (c 0.5, CHCl3) with those reported in the literature for the same compound (Marco et al., 1992). Finally, compound 3 was identified (Figs. S7 and S8) by comparing its 1H NMR and ESI MS data the specific optical rotation value [α]25D = +152.4 (c 0.3, MeOH) with those reported in the literature for salonitenolide (Suchý, Herout & Šorm, 1965; Adekenov et al., 1990).
Antimicrobial activity of the isolated metabolites
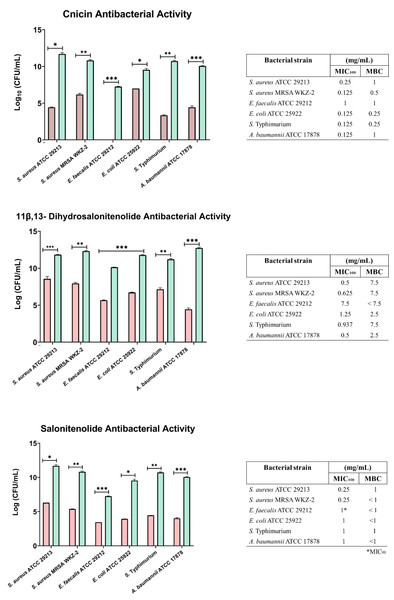
The antibacterial activity of compounds 1–3 was evaluated at different concentrations on E. coli ATCC 25922, S. aureus ATCC 29213, S. aureus MRSA WKZ-2, E. faecalis ATCC 29212, A. baumannii ATCC 17878, and Salmonella enterica subsp. enterica Serovar Typhimurium ATCC 14028 bacterial strains. As shown in Fig. 3, cnicin (compound 1) was found to be the most active compound on all the tested strains. Cnicin was found to be particularly active against Gram-negative bacterial strains E. coli ATCC 25922 and Salmonella enterica subsp. enterica Serovar Typhimurium ATCC 14028 with MIC100 values of 0.125 mg/mL corresponding to minimal bactericidal concentration (MBC) values (Fig. 3). Compound 3, salonitenolide, was found to be effective on all the tested strains even if higher MIC100 and MBC values were detected with respect to cnicin (Fig. 3). Compound 2, 11β,13-dihydrosalonitenolide, showed the lowest antibacterial activity with respect to compounds 1 and 2 with MIC100 values ranging from 0.5 to 7.5 mg/mL (Fig. 3). It has also to be highlighted that, in the case of compound 2, significant differences between MIC100 and MBC values were detected on almost all the strains tested (Fig. 3). In general, MIC100 values obtained for cnicin were found to be similar or even lower than those detected for the CH2Cl2 extract (Table 1 and Fig. 3). The bacterial strain E. faecalis ATCC 29212 was found to be the most resistant to the antimicrobial activity of the tested compounds (Figs. 3 and 4).
Figure 3: Minimal Inhibitory Concentration for compounds 1–3.
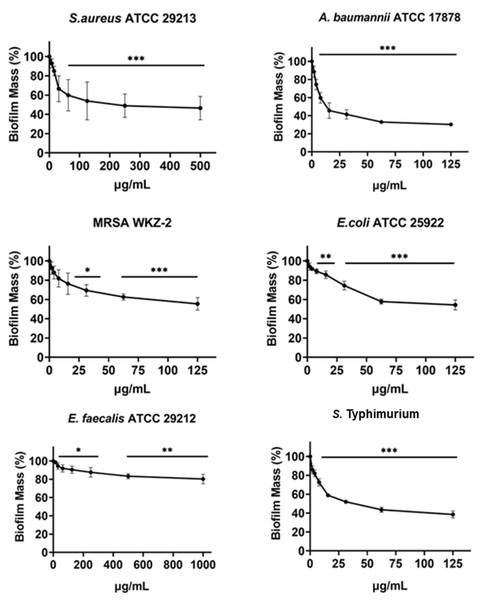
Minimal Inhibitory Concentration MIC100 (mg/mL) and Minimal Bactericidal Concentration MBC mg/mL) values determined for cnicin (compound 1) 11β,13-dihydrosalonitenolide (compound 2) and salonitenolide (compound 3) tested against a panel of Gram-negative and Gram-positive bacterial strains. Control samples are reported as green bars, whereas treated samples are reported as pink bars. Data represents the mean (±standard deviation, SD) of at least three independent experiments. Significant differences were indicated as *p < 0.05, **p < 0.001 or ***p < 0.0001 for treated versus control samples.Figure 4: Antibiofilm activity of increasing concentrations of cnicin (1) evaluated by biomass assays through crystal violet staining on E. faecalis ATCC 29212, A. baumannii ATCC 17878, S. aureus ATCC 29213, Salmonella enterica subsp. enterica Serovar Typhimurium ATCC 14028, S. aureus MRSA WKZ-2, and E. coli ATCC 25922.
Crystal violet was used to stain the formed biofilm and samples were analyzed by using a plate reader measuring the absorbance at 630 nm. The evaluation of antibiofilm activity is employed through biomass assays in which the absorbance value at 630 nm of the treated biofilm is divided by the absorbance value at the same wavelength for the untreated biofilm, in this way the percentage of biofilm mass is indicated. Data represent the mean (±standard deviation, SD) of at least three independent experiments. Each experiment was carried out with triplicate determinations. Significant differences were indicated as *p < 0.05, **p < 0.001 or ***p < 0.0001 for treated versus control samples.In the current study it was found that the isolated cnicin and salonitenolide were endowed with significant antibacterial activity against the tested Gram-negative and Gram-positive bacterial strains (MIC100 = 0.25–1 mg/mL). 11β,13-Dihydrosalonitenolide (2) was found to have a lower antimicrobial activity (MIC100 = 0.5–1.25 mg/mL) in comparison with the other two isolated compounds. Among the tested bacterial strains, E. faecalis ATCC 29212 was found to be the most resistant to the antimicrobial properties of these metabolites.
Evaluation of the antibiofilm activity of cnicin (1)
The antibiofilm activity of cnicin (compound 1) was evaluated by using biomass assays through crystal violet staining on E. faecalis ATCC 29212, S. aureus ATCC 29213, S. aureus MRSA WKZ-2, A. baumannii ATCC 17878, Salmonella enterica subsp. enterica Serovar Typhimurium ATCC 14028 and E. coli ATCC 25922 bacterial strains upon incubation with increasing concentrations (Fig. 4). The biofilm formation was significantly inhibited (about 30–50%) for all the tested strains except for E. faecalis ATCC 29212. The strongest effects on biofilm formation were observed for Salmonella enterica subsp. enterica Serovar Typhimurium ATCC 14028 and A. baumannii ATCC 17878 (Fig. 4) and significant effects were obtained at concentrations of cnicin (1) lower than MIC100 values detected on the same bacterial strains (S. aureus MRSA WKZ-2, S. aureus ATCC 29213, and E. faecalis ATCC 29212 in Fig. 3).
Evaluation of the effects of cnicin (1) on polysaccharides and proteins content in biofilm of tested bacterial strains
Bacterial strains S. aureus ATCC 29213, S. aureus MRSA WKZ-2, E. faecalis ATCC 29212, A. baumannii ATCC 17878, E. coli ATCC 25922, and Salmonella enterica subsp. enterica Serovar Typhimurium ATCC 14028 were selected to analyze the effects of cnicin (1) on proteins and polysaccharides content in biofilm matrix produced by bacterial cells. To do this, Bradford assay and phenol-sulphuric acid method were used to quantify proteins and polysaccharides, respectively (Figs. 5 and 6). It was demonstrated that the treatment with sub-MIC concentrations of compound 1 induces a significant decrease of polysaccharides’ content in all the tested bacterial strains except for E. faecalis ATCC 29212 (Fig. 5). Similar results were obtained when the effects of compound 1 were tested on the protein content of biofilm matrix produced by the same bacterial strains. A significant reduction of biofilm protein content was observed upon treatment with sub-MIC concentrations of compound 1 with more pronounced effects in the case of Gram-negative strains with respect to Gram-positive ones (Fig. 6). Further investigations on compound 1 antibiofilm properties revealed significant effects on biofilm mass when the tested bacterial strains were treated with sub-inhibitory doses of cnicin. In particular, the Gram-negative A. baumannii ATCC 17878 and Salmonella enterica subsp. enterica Serovar Typhimurium ATCC 14028 bacterial strains were found to be the most susceptible strains (Fig. 4).
Figure 5: Quantification of polysaccharides in biofilm matrix produced through 24 h upon incubation with increasing concentrations of cnicin (1) by phenol-sulphuric acid method on E. faecalis ATCC 29212, A. baumannii ATCC 17878, S. aureus ATCC 29213, S. Typhimurium ATCC 14028, S. aureus MRSA WKZ-2, and E. coli ATCC 25922.
Samples absorbance was measured at 482 nm and data represent the mean (±standard deviation, SD) of at least three independent experiments. Each experiment was carried out with triplicate determinations. Significant differences were indicated as *p < 0.05, **p < 0.001 or ***p < 0.0001 for treated versus control samples.Figure 6: Quantification of proteins in biofilm matrix produced through 24 h upon incubation with increasing concentrations of cnicin (1) by Bradford assay in on E. faecalis ATCC 29212, A. baumannii ATCC 17878, S. aureus ATCC 29213, S. Typhimurium ATCC 14028, S. aureus MRSA WKZ-2, and E. coli ATCC 25922.
Samples absorbance was determined at 592 nm and data represent the mean (±standard deviation, SD) of at least three independent experiments. Each experiment was carried out with triplicate determinations. Significant differences were indicated as *p < 0.05, **p < 0.001 or ***p < 0.0001 for treated versus control samples.Discussion
A bio-guided purification of C. hyalolepis organic extract obtained in CH2Cl2 was performed to identify bioactive metabolites responsible for its antimicrobial properties. Through the purification process, fractions with antimicrobial activity were analyzed and it was revealed the presence of three sesquiterpene lactones (STLs) identified as cnicin, 11β,13-dihydrosalonitenolide, and salonitenolide (1–3). C. hyalolepis belongs to the well-known Asteraceae, also named as Compositae family (Ayad & Akkal, 2019). Centaurea plant species were reported to be very rich in STLs and flavonoids metabolites (Ismail et al., 1986). Cnicin (compound 1), one of the compounds identified here, was isolated for the first time in 1960 from Cnicus benedictus (Suchý et al., 1960), a plant that grows in the Mediterranean regions and used for thousands of years to cure liver diseases, anorexia, indigestion problems, ulcers, and swollen fingers (Berger & Sicker, 2009). Cnicin was then isolated from several plants (Suchý et al., 1960; Marco et al., 1992; Adekenov et al., 1990; Ayad & Akkal, 2019; Berger & Sicker, 2009) and analyzed over years to evaluate its beneficial pharmaceutical properties (Rustaiyan, Niknejad & Aynehchi, 1982; Mezache et al., 2010; Belkacem et al., 2014; Lakhal et al., 2010; Kitouni, Benayache & Benayache, 2015; Grafakou et al., 2018; Fernández, Pedro & Polo, 1995). The compound 2, 11β,13-dihydrosalonitenolide, was isolated for the first time from the aerial parts of C. calcitrapa together with cnicin (Marco et al., 1992). Furthermore, it was isolated from the aerial parts of C. alba, C. spinosa and C. pullata (Fernández, Pedro & Polo, 1995; Saroglou et al., 2005; Djeddi, Argyropoulou & Skaltsa, 2008). Salonitenolide (compound 3) was isolated for the first time from C. salonitana in 1965 (Suchý et al., 1960), and then from Ambrosia artemisiifolia (Porter et al., 1970), C. malacitana (Barrero, Sánchez & Arana, 1988), and C. melitensis (Ayad et al., 2012) together with other sesquiterpene lactones, such as cnicin, thus indicating that STLs represent important bioactive compounds of several medicinal plants endowed with anti-inflammatory, anti-diabetic, anti-malarial, anti-proliferative, anti-parasitic, and antibacterial properties (Salazar-Gómez et al., 2020).
The isolated cnicin and salonitenolide were found to be endowed with antibacterial activity against the tested Gram-positive and Gram-negative bacterial strains (MIC100 = 0.25–1 mg/mL). The compound 2, 11β,13-dihydrosalonitenolide, was found to have a lower antimicrobial activity (MIC100 = 0.5–1.25 mg/mL) in comparison with the other two isolated compounds. Among the tested bacterial strains, E. faecalis ATCC 29212 was found to be the most resistant to the antimicrobial properties of these metabolites. Although compounds 1–3 belong to the class of sesquiterpene lactones, key differences in their structural features might be responsible for the detected variations in their antibacterial efficacy (Fig. 2). Moreover, several factors were reported to influence STLs antibacterial properties, such as the compound structure, concentration in solution, hydrophobicity, geometric orientation, the specific bacterial strain tested, and the chemical environment (Schmidt & Heilmann, 2002). By comparing the antimicrobial activity of the purified compounds with that of the total extract from C. hyalolepis, it appears that the isolated pure compounds were endowed with stronger antibacterial properties. This might be correlated to several factors, such as compounds solubility, pH, iron concentration or antagonistic effects exerted by several metabolites acting in a mixture (Beigomi, Biabangard & Rohani, 2021).
Taking into account that cnicin is the strongest antibacterial compound among the three isolated, it is plausible that it mostly contributes to the antibacterial properties of the whole extract. Cnicin is a STL that belongs to the germacranolide group of terpenes (Berger & Sicker, 2009), characterized by an exocyclic α-methylene group in addition to γ-butyrolactone ring (Berger & Sicker, 2009). It was demonstrated that this α-methylene group is responsible for the antimicrobial activity of sesquiterpene lactones in general and of cnicin in particular (Dimkić et al., 2020; Adekenov, 1995). It is worth-noting that cnicin might react through a Michael addition mechanism with nucleophilic groups present in surrounding target molecules, a molecular event that might be at the basis of cnicin antimicrobial activity (Ismail et al., 1986). Based on this, the absence of this structural feature in 11β,13-dihydrosalonitenolide (2) might be responsible for the significantly lower antimicrobial activity of this compound with respect to that of cnicin under the experimental conditions tested. In the literature, it was reported that cnicin interacts with proteins found in bacterial membranes, thus causing cell lysis and death (Dimkić et al., 2020). Cnicin also acts by blocking bacterial cell wall synthesis through the irreversible inhibition of MurA, an enzyme responsible for the catalysis of the first step of the synthesis of peptidoglycans (Bachelier, Mayer & Klein, 2006).
Interestingly, cnicin was also found to be endowed with significant antibiofilm properties. Furthermore, a remarkable decrease in the biofilm protein and exopolysaccharide content was evidenced upon treatment with cnicin (Figs. 5 and 6), thus further highlighting the significant antibiofilm properties of compound 1. The EPS (extracellular polymeric substance) matrix develops a three-dimensional network that shields bacterial cells from antimicrobials and host immune system defense strategies (Steenackers et al., 2012). Additionally, the EPS matrix promotes surface adhesion and colonization. Hence, its inhibition or disruption could be considered a key event to prevent or to eradicate biofilms (Palomares-Navarro et al., 2023). Previous studies have revealed that the antibiofilm properties of plant-derived compounds, such as phenolics and terpenes, might be correlated to their ability to interfere with EPS production (Ortega-Ramirez et al., 2020; Keelara, Thakur & Patel, 2016; Hui et al., 2016; Gutierrez-Pacheco et al., 2018). Moreover, some studies suggested the ability of cnicin to interfere with quorum-sensing (QS), a key step leading to biofilm formation (Hentzer et al., 2002). The currently achieved results provide evidence that cnicin is able to prevent biofilm formation, thus opening interesting perspectives to the treatment of antimicrobial resistance (AMR) development.
Conclusion
To the best of our knowledge, this is the first study aimed at identifying metabolites responsible for the antibacterial activity of C. hyalolepis collected in Palestine. To do this, the plant dichloromethane (CH2Cl2) organic extract was used as starting point to perform a bio-guided purification of active metabolites. Three sesquiterpene lactones (STLs) were isolated and identified as cnicin, 11β,13-dihydrosalonitenolide, and salonitenolide (compounds 1–3, respectively). Cnicin was found to be endowed with the strongest antimicrobial activity towards Gram-negative and Gram-positive bacterial strains. Moreover, cnicin also resulted to be a potent antibiofilm agent, thus opening the way to further studies aimed at deeply characterizing its biological properties and its applicability in biomedical field to counteract multidrug resistant infections.